For the 701 mammals that occur in Brazil (ca. 13% of extant mammals worldwide), we attributed ES to each deliverer one based on their functional traits, known trophic interactions, expert knowledge, and by overlaying IUCN's distribution maps of the species. The ES term encompasses both ecosystem processes and services, offering a broader perspective that enables us to investigate the potential benefits of mammals to people. We assumed that species that are currently listed as threatened in the IUCN most likely lost their ES role. We found that 82% of Brazilian mammals (575 spp.) deliver at least one of the 11 ES identified. Cultural services were associated with the greatest number of deliverer species, while carrion control, ecosystem engineering, and rodent control were delivered by fewer species. Although only 24% of the species analyzed here are endemic to Brazil, 75% of them deliver at least one ES. The ES they delivered are also disproportionately at risk, as 57% of the species considered to have lost their ES role are endemic. Ecosystem service provision does not merely mirror the spatial patterns of mammal richness in general, as we identified hotspots for each service across Brazil, including its seascapes. Our comprehensive assessment provides a clear overview of the myriad of services delivered by Brazilian mammals, including taxonomic and spatial explicit information for each service. Identifying and mapping species and their services can contribute to more effective management and conservation programs focused on optimizing the supply of ES and conserving biodiversity.
Biodiversity is involved in a number of ecosystem functions and processes which refer to the biophysical, chemical, and biological activities and interactions that occur within an ecosystem, driving the flow of energy, nutrients, and materials (IPBES, 2019). These processes can sustain or be considered as ecosystem services (ES) or nature’s contributions to people, as they are essential for human life and well-being (Harrison et al., 2014; IPBES, 2019; Manes et al., 2022). Ecosystem services can be classified in three major types: (i) provision, which includes nutritional, non-nutritional and energy products, (ii) regulation, which are ways in which systems can mediate or moderate the environment, and (iii) cultural, which are non-material benefits that affect people’s physical and mental state (sensuHaines-Young and Potschin, 2018). Species can deliver ES directly, by pollinating crops for example (a.k.a. regulatory service), or indirectly by pollinating native plants, which in turn will deliver recreational activities (a.k.a. final cultural service) (Haines-Young and Potschin, 2018; Keyes et al., 2021). Biodiversity can also provide services indirectly through fundamental ecosystem processes, such as nutrient cycling, primary productivity, and habitat, which are known as supporting services (the fourth type of ES; sensuMEA, 2005).
Attributing ES to biodiversity, however, is scientifically challenging. Ecosystem services are often attributed to biodiversity at the ecosystem-level because quantifying it at the species-level can be difficult (Balvanera et al., 2014; Harrison et al., 2014; Keyes et al., 2021; Kremen, 2005; Ramos et al., 2020). Even at the species level, most studies focus on species with direct economic interest (Ferreira et al., 2021; Harrison et al., 2014), and little is known about the functions and processes that are delivered or supported by most species (known as Raunkiæran (Hortal et al., 2015) or Eltonian shortfall (Rosado et al., 2016)). This is particularly true in the tropics, where biodiversity is enormous, and so are its associated knowledge shortfalls (Hortal et al., 2015; Pimm et al., 2010). However, understanding and identifying each species provides each ES is fundamental to adopting efficient strategies for species and ES conservation and guiding public policies.
Being able to attribute ES to species is very important for their conservation and management, especially considering that species are disappearing at great speed. Although we have been able to estimate to some extent the current rates of species extinction (IPBES, 2019), we are quite clueless in terms of its consequences to associated ES, in part because these services are seldom associated with individual species. The Millennium Ecosystem Assessment (MEA, 2005), for example, estimated that 60% of the ESs evaluated in the assessment were being degraded or used unsustainably. Although the assessment clearly states that changes in species affect ecosystem processes, the estimated trends in ESs were rarely tied to the loss of species itself, but to environmental degradation in general, except for some provisioning services such as those associated to medicinal plants or fisheries. This shortage of a direct attribution of ESs to species remains. Even when studies explicitly acknowledge biodiversity's role in ES provisioning, they seldom attribute such contributions to individual species' identities (Haines-Young and Potschin, 2010; Harrison et al., 2014; Manes et al., 2022). Considering the recent advances in ES studies in Brazil, for example, about 80% of them used native vegetation as a proxy of biodiversity without considering species' contributions to the services evaluated (Pires et al., 2018). Despite being a traditional approach in ES studies, it fails to contribute to the implementation and designing of strategies dedicated to promoting species conservation.
In the rare instances when ES are directly attributed to species, these are often plants, used as a food, medicine or timber (Ferreira et al., 2021; Harrison et al., 2014; Schulp et al., 2014). Other taxonomic groups, however, are also likely to be tied to ESs. Mammals, for example, are an extensively studied taxonomic group (IUCN, 2020; Soria et al., 2021; Wilman et al., 2014), suggested to be tied to delivering and supporting several ESs, such as cultural and aesthetic, pollination, pest control, soil fertility, and seed dispersal (Civantos et al., 2012; Kremen, 2005; Lacher et al., 2019). There are several mammals that are economically important for the services they deliver, such as pollinator and insectivorous bats, which support agricultural systems (Aguiar et al., 2021; Kunz et al., 2011; Ramírez-Fráncel et al., 2022), and charismatic species, such as jaguars and giant anteaters, which support ecotourism activities (Möcklinghoff et al., 2014; Tortato and Izzo, 2017). Considering the wide societal interest in mammals and the availability of information on their traits, feeding, and foraging behaviors – which can be used to tie species to ESs (Bello et al., 2010; Díaz and Cabido, 2001; Harrison et al., 2014) – they are perfect candidates for a systematic assessment of individual species’ provision of ESs.
Here, we carried out this exercise for around 700 mammal species native to Brazil, a megadiverse country that is quickly building knowledge on the ESs delivered and supported by its biodiversity (BPBES, 2018; Pires et al., 2018). This study has three objectives: (i) creating a comprehensive database on the ESs delivered directly and indirectly by Brazilian mammals, (ii) describing mammal diversity patterns associated with each ES, and (iii) mapping the distribution of ES associated with mammals across Brazil.
MethodsWe compiled the list of 701 species of mammals native to Brazil (according to Jenkins et al., 2015; IUCN, 2022), not considering subspecies. Of those, 169 (24%) are endemic mammals to Brazil. Because attributing ESs to individual species can be challenging, studies often use species’ traits, instead of direct evidence, to determine the ES delivered and supported by them (Bello et al., 2010; Díaz and Cabido, 2001; Harrison et al., 2014; Truchy et al., 2015). Species’ traits are associated with their ecological function and, consequently, to ecosystem processes and services (Bello et al., 2010; Díaz and Cabido, 2001; Harrison et al., 2014). Thus, we used the traits of Brazilian mammals compiled from online databases (IUCN, 2022; Myers et al., 2020), data papers and books (see Table S1). The databases were used as a first assessment of likely ES associated to species, which was then scrutinized by the experts in different taxonomic orders in the author team. When there was no information or the information obtained was mixed, we (i) opted for the most reliable and current database, (ii) considered experts’ knowledge, and (iii) actively searched for the information in other published sources (Table S1). We compiled information on order or suborder, family, diet, body mass, foraging stratum (terrestrial, marine, freshwater, and fossorial species), dispersal ability, home range size, and threat state (Table S1).
Based on existing knowledge on the ES delivered or supported by vertebrates (e.g., Sekercioglu, 2006; Lacher et al., 2019), we defined 11 ecosystem processes and services that could be associated to mammals: (i) relational service delivered by charismatic species and (ii) ecotourism; (iii) pollination, (iv) seed dispersal, (v) pest and disease control, (vi) rodent control and (vii) disease sentinelling; (viii) carrion control, (ix) nutrient transporting, (x) top-down regulation, and (xi) ecosystem engineering. We adhered to the classification outlined in the 2005 Millennium Ecosystem Assessment (MEA), which, though old, still enjoys broad acceptance and extensive use. More recent classifications, such as IPBES’ Nature’s Contribution to People, offer different typologies based on recent discussions, some of which even question the relevance of the ecosystem service concept in itself (Pires et al., 2020; Díaz et al., 2018). To ensure the broadest accessibility and usability of our results, we chose to adopt the more inclusive perspective advocated by the MEA, which classifies ecosystem processes as supporting services. Therefore, it's important to note that when we use the term "ecosystem services" (ES) here, it encompasses both ecosystem processes and services, aiming for maximum comprehensiveness in our assessment of the potential benefits of mammals to people. In our detailed description of each ecosystem service in the Supporting Information, we discuss whether it can be considered a service, a process or both. Importantly, our dataset of species and their associated ecosystem services are flexible enough to accommodate various conceptual and classificatory approaches, allowing users to focus on services, processes, or even combine different ecosystem services as needed.
The first two ES of the list are classified as cultural and aesthetic services and the others as regulation services (or supporting services, or even ecosystem processes). The well-known mammalian provision service associated with game meat (Haines-Young and Potschin, 2018) was not considered here because hunting of wildlife is illegal in Brazil, except for subsistence hunting by indigenous peoples (see Supporting Information for more detail; Brasil, 1967, 1973), and the hunting of the invasive crossbreed between the domestic pig and the European wild boar (IBAMA, 2013). For this study, we considered native mammals in wild state, not including services delivered by their farmed counterparts (e.g., capybaras, Hydrochoerus hydrochaeris (Nogueira-Filho and da Cunha Nogueira, 2018)). In addition, we did not consider services delivered by native species outside their native range. For example, we considered the common marmoset, Callithrix jacchus, as a charismatic species, but ignoring areas of its distribution where it became invasive (Zaluar et al., 2022). See the Supporting Information for further details on the methods used to define and choose each ES.
The Eltonian and Raunkiaeran shortfalls (i.e. the lack of knowledge about species’ interactions and species’ traits and their ecological functions, respectively) are especially pronounced in the Global South (Hortal et al., 2015). Thus, whenever possible, we extrapolated from known species-ES relationships. For example, all species that are nectarivores are also potential pollinators. We also made extrapolations based on phylogenetic relationships; for example, if species in a genus were known to be nectarivores, we considered its congeners without information on diet to also be nectarivores (and thus, pollinators). Finally, we also extrapolated based on traits that are associated with functions; for example, large-wing bats are known to be efficient at catching insects (and thus, do pest control), while small-wing bats are more likely to be pollinators (Kunz et al., 2011).
Abundant species tend to contribute more than rare species to ESs (Díaz et al., 2011; Winfree et al., 2015), except for species that deliver cultural (relational and ecotourism) services. Therefore, species classified as extinct in the wild, threatened (critically endangered and endangered) or data deficient by the IUCN Red List of Threatened Species (https://www.iucnredlist.org/) were considered to have lost their ES role, except for cultural services.
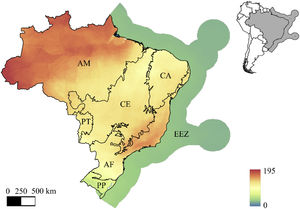
To understand the spatial distribution of ESs provided by mammals in Brazil, we used the distribution map by IUCN for all species that provide at least one ES (IUCN, 2018). To map each of the 11 ES evaluated here, we overlapped the distribution maps of all species that provide each ES, producing a species richness estimation for the ES. Ecosystem service maps were overlaid with the map of Brazil’s six phytogeographic domains (called “biomes”, hereafter) and economic exclusive zone (Fig. 1). These regions are widely recognized throughout Brazil (e.g., IBGE, 2023; MapBiomas, 2021). All analyses of this study were performed in R version 4.0.0 (R Core Team, 2020).
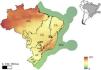
Mammal richness in the Brazilian phytophysiognomic domains (a.k.a. biomes in Brazil) and economic exclusive zone. AM = Amazon, CE = Cerrado, CA = Caatinga, PT = Pantanal, AF = Atlantic Forest, PP = Pampa, and EEZ = Brazilian Economic Exclusive Zone. The color ramp represents the richness of all Brazilian mammals (data from Jenkins et al., 2013, Jenkins and Van Houtan 2016).
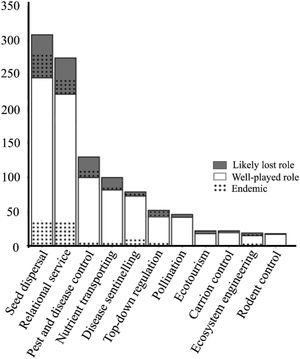
Species that did not deliver any ES evaluated were almost exclusively from the order Rodentia (118 species, including the two extinct rodents, Juscelinomys candango and Noronhomys vespuccii), plus four Didelphimorphia and four Chiroptera species (Table S1). No species delivered more than 7 ES at once (Table S1). Armadillos (genera Dasypus, Tolypeutes, Cabassous, and Euphractus) were the only group identified as a deliverer of most ES (6 and 7 ES).
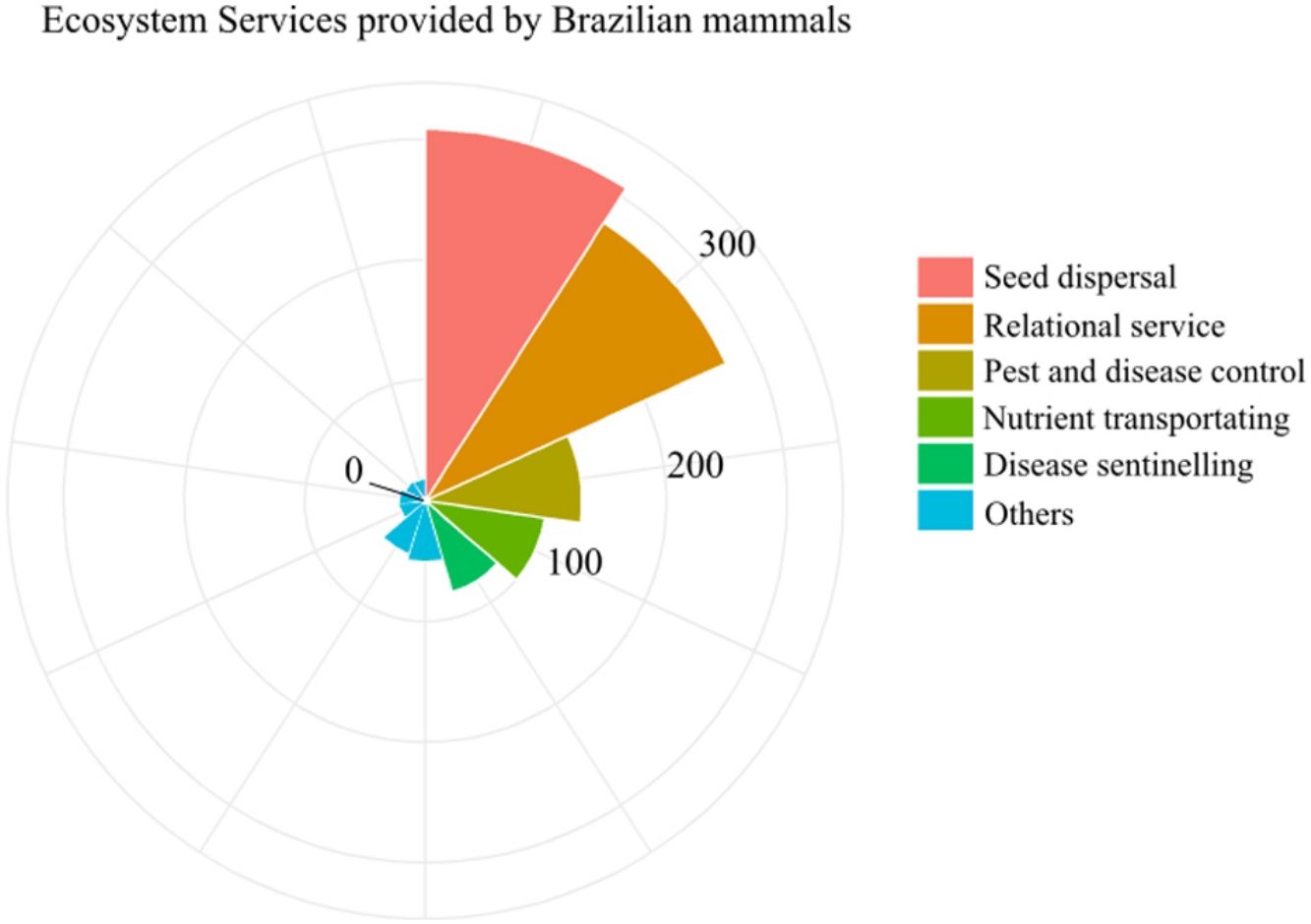
The ESs and processes delivered by the greatest number of species were seed dispersal and relational service (308 and 274 species, respectively). About 138 species identified as performing ES were considered to have lost their role for being threatened or data deficient (Fig. 2, Table S1). This represents one fifth of the mammal species performing ES in the country. The ES of ecotourism, carrion control, ecosystem engineering, and rodent control were the least represented, being delivered by only about 20 species each (Fig. 2). Although only 24% of Brazilian mammals are endemic, they played a large role in ES provisioning, with 75% of them delivering at least one ES, especially seed dispersal, and cultural service (Fig. 2). At the same time, the ES they deliver are disproportionately at risk, as 57% of the mammal species that have lost their ES role are endemic (Table S1).
Number of Brazilian mammal species delivering each of the 11 ecosystem services (ES) assessed. “Well-played role” refers to species considered to fulfill their ES role for being common, while “likely lost role” refers to species considered to have lost their ES role for being extinct, threatened or data deficient. Of these, the proportion of species that are endemic to Brazil is represented with a dotted pattern.
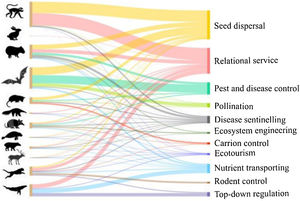
Most species delivering ES belonged to the orders Primates, Rodentia, Chiroptera, Carnivora, and the suborder Cetacea (Fig. 3), which is not surprising, as these are the ones with the greatest number of species in Brazil (Figure S1). At the same time, although Rodentia has the greatest number of species in the country (233 species, Figure S1), 51% of them did not deliver any of the ES considered here (Table S1). Also expectedly, the orders and suborders with low number of species in Brazil delivered fewer services (Artiodactyla, Pilosa, Lagomorpha, Sirenia, and Perissodactyla), except for Cingulata, where all 10 species delivered between 5 and 7 services each (Figure S1, Table S1).
Number of species in each order or suborder delivering each of the 11 ecosystem services assessed. Lines connect orders or suborders to the services they deliver, with line thickness indicating number of species delivering the service. From top to bottom, the orders or suborder indicated by their silhouettes are Primates, Lagomorpha, Rodentia, Chiroptera, Didelphimorphia, Pilosa, Cingulata, Sirenia, Perissodactyla, Artiodactyla, Carnivora, and Cetacea.
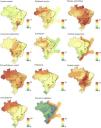
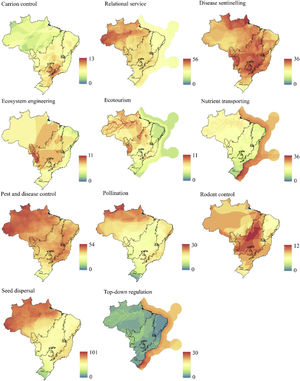
The richness of ES was not homogeneously distributed across the country and its biomes (Figs. 1 and 4). Although in Brazil the highest richness of mammals is found in the Amazon and the Atlantic Forest (Fig. 1), this pattern was not necessarily followed by all ESs delivered by Brazilian mammals (Fig. 4). The Amazon showed high species richness for most ESs, either across most of its area (i.e., ecotourism, relational service, disease sentinelling, nutrient transporting, and pest and disease control), or in part of it (i.e., ecosystem engineering, pollination, rodent control, and seed dispersal), being poor in top-down control and carrion control deliverers (Fig. 4). In the Atlantic Forest, on the other hand, despite having very high mammal richness, there were fewer ESs deliverers. The Atlantic Forest was particularly important for pollination services, and some relevance for services of disease sentinelling, pest and disease control, and carrion control (Fig. 4). The Pantanal was clearly a hotspot for ecotourism, but also disease sentinelling, ecosystem engineering, and rodent control (especially in the south). The Cerrado was an important hotspot for rodent control and disease sentinelling but had also high richness of ecosystem engineering and pest and disease control deliverers (Fig. 4). The Caatinga only showed high richness for disease sentinelling and pest and disease control deliverers, as most of terrestrial biomes, and the Pampa showed low species richness for all services (Fig. 4). The Brazilian Exclusive Economic Zone was an important hotspot for top-down regulation and nutrient transporting. Although the disease sentinelling, pest and disease control, and carrion control services showed some areas with higher species richness, these areas were not restricted to specific biomes, being either high across most of the country (disease sentinelling, pest and disease control), or concentrated in an area among many biomes (carrion control).
DiscussionOur analysis provides a clear overview of the multiple services delivered by different groups of mammals and highlights the potential important contributions of Brazilian biodiversity to people, including the remarkable role of the species that are endemic to the country. Most importantly, we show that service provisioning does not simply reflect spatial patterns of species richness in general, but that it is possible to identify hotspots for each service across Brazil, including in its seascapes. The identification of species contributions to people is a first step towards the recognition of their social-economic importance, especially in a country with such worldwide-recognized natural capital, like Brazil.
Each Brazilian biome has different degrees of loss of vegetation cover, area within protected areas, and often also different environmental protection rules (Brasil, 2000; Câmara et al., 2015; IPCC, 2021; PBMC, 2013; Soterroni et al., 2018). The Amazon, for example, still holds ca. 80% of its original forest cover, and has the largest proportion (ca. 26%) of its remaining forests within protected areas, being currently the least vulnerable Brazilian biome (Diele Viegas et al., 2022; Jenkins et al., 2013; MapBiomas, 2021). This is good news, given that we found high species richness for most ESs in the Amazon. The Atlantic Forest and Cerrado savannas, on the other hand, are Brazil’s two biodiversity hotspots, combining heavy loss of forest cover (>80%) with equally high rates of endemism (Myers et al., 2000), and only ca. 8% of their remaining forests within protected areas (Jenkins et al., 2013). These two biomes show great species richness for half of the ESs, most related with disease controls (e.g., disease sentinelling, pest and disease control, rodent control, and carrion control), which is essential for human health, especially in the Atlantic Forest, which is the most developed and populated region in the country, and a hotspot for emerging infectious diseases at the global level (Allen et al., 2017; Rezende et al., 2018).
It is important to remember that ESs are services that are delivered to people. Thus, in regions with low human population, such as the Amazon, fewer people will benefit from ESs, even if it is a hotspot for many ESs. In the Atlantic Forest, on the other hand, the high human population density means that the few ESs that are being delivered will benefit many more people. Especially in areas with lower concentrations of ES but higher populations, public policies and management strategies must prioritize maximizing access to the ESs offered, for example, through explicit spatial planning, also mapping the ES chain supply-flow-demand (see Metzger et al., 2021). At the same time, ESs may benefit people that are away from the region where they are produced. This is the case, for example, of the climatic and water cycle regulation provided by the Amazon Forest, which benefits people in different regions in South America (Strand et al., 2018). That might not be the case, however, for the ESs delivered by individual animal species.
Interestingly, the small region that shows high species richness for carrion control is in the ecotone between the Atlantic Forest and Cerrado. Carrion control increases the energy flows in food webs, limits the spread of diseases, removes carcasses from the field, and indirectly regulates greenhouse emissions (DeVault et al., 2003; Morales-Reyes et al., 2015; Olson et al., 2012). Another relevant biome for ES in Brazil is the Pantanal, one of the World’s largest wetlands, with high species richness but low endemism, with only ca. 3% of their remaining forests inside protected areas, but a number of Ramsar Convention Wetlands of International Importance sites (Jenkins et al., 2013; Ribeiro et al., 2020). We found the Pantanal to be extremely relevant particularly to ecotourism services. The region is known for its abundant and highly visible wildlife and is indeed considered to be the most important areas for wildlife tourism in Latin America, comparable to well-known parks in African savannas (Tortato et al., 2017).
Finally, the Brazilian Exclusive Economic Zone (EEZ), which includes ∼25% of its waters within protected areas (but see Silva, 2019), was the most important biome for nutrient transporting, a service delivered by aquatic mammals. This service is extremely important for the fertilization of natural and anthropized environments, especially by transferring nutrients from high productive (e.g., pelagic) to low productive waters (e.g., deep ocean) (Doughty et al., 2013; Roman et al., 2014; Sekercioglu, 2006). The EEZ also shows the highest species richness for the top-down regulation, especially in the south, where waters are more productive. Top predators consume large amounts of biomass, providing an important top-down process, which stabilizes trophic dynamics, and leading to higher species richness via competitive coexistence (Estes et al., 2016; Ives and Dobson, 1987; Wallach et al., 2015).
Most biomes show high richness of disease sentinelling species and those that perform pest and disease control, indicating that these are the least vulnerable ESs in Brazil. Many mammals work as species sentinels as they participate in the cycle of zoonotic diseases that affect humans, being useful in determining pathogen distribution, abundance, and understanding disease ecology (Plowright et al., 2017; Winck et al., 2022). The mammals that work best for early detection of epidemics are those that interact with wildlife, humans, and domestic animals or livestock (Plowright et al., 2017; Winck et al., 2022). Therefore, monitoring the prevalence of pathogens in mammal populations is extremely important to predict the risk of disease outbreaks in humans (Rodrigues et al., 2020). Preventing outbreaks of pathogens by monitoring these mammals should be a priority for public policies in Brazil, as they are cost effective and can save human lives (Winck et al., 2022). Regulatory species that deliver pest and disease control service, on the other hand, contribute to increasing crop yields and food security, reducing the need for pesticides, improving ecosystem integrity and human health (Maas et al., 2013; Wanger et al., 2014).
Surprisingly, our analysis reveals that the top deliverers of ESs in Brazil are some armadillo species, which deliver up to seven services each. Armadillos are burrowing mammals well-known for their roles as ecosystem engineers and bioturbators (considered here as nutrient transportation service), moving and mixing large quantities of soil and sediment to the surface, and creating new habitats (Haussmann, 2017). These new habitats can be a refuge for many other animals (e.g., beetles, social wasps, lizards, snakes, frogs, and small mammals), increasing abundance and richness of invertebrates and vertebrates, being important for the whole ecosystem community (Rodrigues et al., 2020). These animals also have a great potential to be disease sentinels as they are reservoir hosts of important pathogens (e.g., Trypanosoma cruzi, the pathogen of the Chagas disease, Yeo et al., 2005). Unfortunately, Brazil does not have a well-established biomonitoring program to track the prevalence of pathogens in armadillos (Rodrigues et al., 2020). In addition, armadillos are intensively hunted in Brazil (Rodrigues et al., 2020). At the other extreme, the Lagomorpha (rabbits) is the group of mammals that delivers the fewest ESs in Brazil. There is a low diversity of Lagomorpha in Brazil, which were considered charismatic species, and thus, deliverers of relational services. There is evidence of seed dispersal for some rabbits in other countries, but not for Brazilian species (Godó et al., 2022).
Most mammals in Brazil deliver seed dispersal and relational services (>270 species each). The high redundancy on seed dispersal provision safeguards this essential ecological process, at least for plants that are dispersed by mammals. Zoochory is especially prevalent in the tropical forests, with about 50 %–90 % of plants being dispersed by animals (Almeida-Neto et al., 2008; Fleming et al., 1987). Mammals have also been widely associated with relational services, especially because of the high number of charismatic species, and this was captured in our study, with 274 (c.a. 40% of all Brazilian mammals) species. Despite the large number of charismatic species, which translates into a relatively high level of relational services across the country, a much lower number of species delivers ecotourism services. We found only 15 terrestrial mammals and seven aquatic mammals (freshwater Amazonian and marine large mammals) that are mentioned in ecotourism activities in Brazil, and only few of them have a direct ecotourism activity devoted to them, such as Panthera onca (jaguar) in Pantanal and Inia geoffrensis (pink dolphin) in the Amazon (D’Cruze et al., 2017; Tortato and Izzo, 2017). Most of species mentioned in ecotourism activities have side roles, as exemplified by Nasua nasua (coati) in the Iguaçu falls, which is pointed out as one of the attractions, although the highlight of the tour is clearly the falls themselves (https://www.visitefoz.com.br). The low number of species delivering ecotourism services may be due to the fact that most charismatic mammals are difficult to see in the wild in Brazil. Still, there seems to be a high untapped potential for mammal-based ecotourism in Brazil, which could anchor conservation policies and initiatives in the country (see Simberloff, 1998).
The low redundancy (i.e., species richness) in important ES, either across the country or in part of it, highlights the need to conserve the individual species delivering them. We identified important ES and processes supported by very few species across the country, showing low redundancy and, thus, a high vulnerability for their provision – especially for services that are almost exclusively associated with mammals, such as ecosystem engineering (Coggan et al., 2018; Romero et al., 2015). Ecosystem engineering, carrion elimination, and rodent control are services delivered by less than 20 species in the country. Clearly, some ESs that have low redundancy within mammals, such as rodent control and carrion elimination, can also be delivered by other vertebrates, such as birds (e.g., raptors and vultures) or reptiles (snakes) (DeVault et al., 2003), although mammals play a more relevant role. Many rodents are known as reservoirs of human pathogens (Civitello et al., 2015), and thus, controlling their population growth is important for human health. In addition, rodent predators can maintain high rodent diversity by avoiding competitive exclusion, which is essential to reduce the dominance of competent rodent hosts and the associated transmission of diseases (Civitello et al., 2015), a process known as “dilution effect” (Ostfeld and LoGiudice, 2003; Schmidt and Ostfeld, 2001).
Pollination also has low species redundancy, being delivered by 51 species (7% of all analyzed mammals), but with an estimated maximum of 30 per pixel in some areas of the Amazon. Pollination is an important process for plant reproduction and fruit and seed formation, recognized as a regulatory ES that ensures crop yields and food security (Wolowski et al., 2019). Although most economically important crops are pollinated by invertebrates, some are specialized in mammal pollinators, such as the pequizeiro Caryocar brasiliensis and C. villosum, that produces a fruit widely consumed in the Brazilian Cerrado and are pollinated by bats (e.g., Anoura geoffroyi and Glossophaga soricina, Wolowski et al., 2019). Thus, the vulnerability of this service delivered by mammals could trigger shortage of these products.
It is worth mentioning that here we considered the IUCN range maps to derive species richness and its ESs. However, species abundance is a crucial factor to a species effectively delivering an ES (e.g., Gaston et al., 2018). If the population density is too low, species may be only marginally (if at all) contributing to providing ESs. On the other hand, large populations may have a higher contribution to ESs. Thus, species abundance may be more important than species presence in effectively delivering ES (Gaston et al., 2018; Dainese et al., 2019). However, here we used species richness as a proxy for ecosystem services provided by mammal species. Therefore, even if a species is locally rare, if the richness of species providing that service is high, we can assume that the ecosystem service is being delivered despite having no information on species’ abundance.
It is important to consider the ES delivered by these species at the local and regional scale should be considered in conservation policies. For example, our study helps identify beneficiaries of the services offered by these species and alert for the need of these species’ conservation. Some of the species highlighted here provide unique and strategic ES and should be the focus of local and regional species conservation plans. Under Ecosystem Based Management (EBM) approaches, the ESs and species that deliver them should be included in Protected Areas management plans and used to monitor the current benefits provided.
Tropical regions concentrate most of the global biodiversity but have the highest biodiversity knowledge gaps (Hortal et al., 2015), especially when it comes to the roles played by this rich biodiversity (Rosado et al., 2016). Here, we provide the first comprehensive assessment of all estimated ES delivered by mammals at the country level. Given Brazil’s continental scale and diverse mammalian fauna (Jenkins et al., 2015), this represents an assessment of 13% of the ca. 5,400 extant mammals worldwide. Knowledge of their role in ESs tackles this important knowledge gap in South America, especially considering that only 24% of the Brazilian mammals are endemic, and thus, most species evaluated here also occur in other countries, likely delivering the same ES. Knowing and quantifying the ESs delivered by species can also contribute to more effective management, focused on the dual goal of optimizing the delivery of ESs and supporting biodiversity conservation, which is essential for a sustainable future (Harrison et al., 2014; Pörtner et al., 2021).
Conflict of interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
This study was inspired by LO and WT’s ongoing work on the ecosystem services provided by European vertebrates. MMV developed this work during a sabbatical year at the Laboratoire d’Ecologie Alpine (LECA), Université Grenoble Alpes, France, with support from CNPq (grant 202284/2020-4, 304908/2021-5). MMV and RT also received support from the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ, grant E-26/202.647/2019 and E-26/ 200.238/2023). LT received a fellowship from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grant 381795/2021-7), and from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, grant #2022/07192-1). This paper was developed in the context of the Services Delivered by the Brazilian Fauna Project, conceived and coordinated by MMV; the Brazilian Research Network on Climate Change, supported by FINEP (grant 01.13.0353-00); and the National Institute for Science and Technology (INCT) in Ecology, Evolution and Biodiversity Conservation, supported by MCTIC/CNPq (grant 465610/2014-5) and FAPEG (grant 201810267000023). APFP thanks for the support of Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ, grants E-26/210.164/2019, E-26/211.988/2021, E-26/201.454/2022) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grant 423057/2021-9). MMW thanks the Financiadora de Estudos e Projetos (FINEP) for financial support (MCT/FINEP/CT-INFRA-CAMPI REGIONAIS-01/2010).