It is now clear that dry forest regeneration relies on a set of regeneration mechanisms such as seed rain, seed and seedling banks, and resprouts that operate simultaneously, but their relative contribution and drivers remain poorly understood. In this paper, we examined the role of seed rain, seed soil bank, seedling, sapling, and resprout assemblages in the regeneration of a Caatinga dry forest in northeast Brazil. We also examined how these regeneration mechanisms respond to chronic disturbances, rainfall, soil fertility, and light in 18 plots located in old-growth forest stands. A total of 5239 seeds from seed rain, 932 seeds in the soil seed bank, 158 seedlings, 85 saplings and 416 resprouts were recorded. Overall regeneration mechanisms provided low-density and impoverished assemblages when examined isolated. Soil seed bank and seed rain provided up to 423 and 35.5 seeds per m2, respectively. Seedling and sapling assemblages were dominated by a small set of species (mostly Jatropha mutabilis, Pityrocapa moliliformis, and Trischidium molle), which were both frequent and accounted for most of the individuals. Resprouting was the most important mechanism, accounting for 63.1% of all individuals in the seedling, sapling, and resprout assemblages. Chronic disturbances were identified as key drivers, negatively affecting several community-level attributes of seed rain, soil seed bank, seedlings, and resprouts. Soil fertility and leaf area index (i.e., light availability) were negatively related to seedling and sapling abundances, respectively. Furthermore, the effect of environmental factors on regeneration was more pronounced in disturbed areas. Species composition of saplings and resprouts were associated with chronic disturbances and environmental factors. Our results suggest that, in human-modified landscapes, Caatinga forest regeneration relies primarily on resprouting, while other mechanisms play a minor role in plant species recruitment and maintenance along gradients of disturbances and environmental factors. Given the predicted increase in aridity associated with increased human disturbance, the Caatinga dry forest may experience shifts in forest structure and regeneration patterns. We refer to an absolute prevalence of vegetative propagation leading to a low-statured forest dominated by multi-stem individuals, with unknown effects on long-term forest dynamics and resilience, including community-level homogenization.
The survival and persistence of forest ecosystems facing of global changes over the years are the result of several processes such as forest regeneration (Anderson-Teixeira et al., 2013; Chazdon, 2008). Tropical forest regeneration refers to the process by which a forest regrows/recovers after experiencing natural or anthropogenic disturbances (McDonald et al., 2010; Vieira and Scariot, 2006). In this context, the regeneration is primarily driven by a set of key mechanisms, namely seed rain, soil seed bank, seedling and sapling assemblages, and resprouts from adult individuals Chazdon, 2003; Chazdon, 2012; Cury et al., 2020). In fact, these mechanisms play pivotal roles in (1) forest regeneration and dynamics, (2) plant assemblage attributes, and (3) the physical structure and biomass of the forest. Forest regeneration mechanisms respond differently to numerous drivers, including seed production, water and nutrient availability, microclimate conditions, and seed disperser availability (Grime, 1977; Pickett et al., 1999). For instance, seed predation, competition from grasses, competition from exotic or invasive plants, drastic environmental conditions, and soil compaction and degradation pose threats to the recruitment of regenerants and the overall forest regeneration (Aide and Cavelier, 1994; Alvarez-Aquino et al., 2004; Derroire et al., 2016; Nepstad et al., 1996; Quesada et al., 2009). Furthermore, intense human land use, characterized by the extensive removal of biomass, has received considerable attention due to its significant impact on the recovery process (Marinho et al., 2016; Poorter et al., 2016; Sakio, 1997). However, there is an urgent need to quantify and predict the effects of less intense, persistent, and recurrent disturbances such as chronic anthropogenic disturbances (sensu Singh, 1998) on regeneration mechanisms.
Although disturbances and environmental factors (rainfall, soil fertility, and light availability) significantly influence ecological processes such as forest regeneration, there is a lack of comprehensive studies that examine the contribution of each regeneration mechanism to the forest recovery process. Existing research has mainly focused on describing, quantifying, and assessing the effect of disturbances and/or abiotic factors on one or two regeneration mechanisms (Holz et al., 2009; Lieberman and Li, 1992; Nepstad et al., 1996; Vieira et al., 2006; Chaturvedi et al., 2017; Marinho et al., 2016). Studies that quantify all the regeneration mechanisms, determine their relative contribution to the recovery process, and assess their relationships with environmental factors are very scarce (Ceccon et al., 2006; Kennard et al., 2002; Murphy and Lugo, 1986; Vieira et al., 2007).
Chronic anthropogenic disturbance (hereafter chronic disturbance) refers to the removal of forest biomass through different activities, such as firewood collection, non-timber forest products exploitation, livestock raising, and hunting, among other forms of natural resource use (Singh, 1998). In combination with slash-and-burn agriculture (i.e. forest degradation sensu Ghazoul et al., 2015), this type of disturbance can lead to arrested succession, thus limiting or completely halting ecological processes essential for forest regeneration (Ghazoul et al., 2015). Chronic disturbance is particularly common in dry forests inhabited by forest-dependent people (McLaren and McDonald, 2003; Ribeiro et al., 2015), where their negative impact on forest integrity, dynamics, and regeneration has been reported (Antongiovanni et al., 2020; Barros et al., 2021; Ramos et al., 2023). Chronic disturbance negatively affects seed rain, soil seed bank, and seedling recruitment in dry forests (Bezerra et al., 2023a, 2022; Luna-Nieves et al., 2019; Trindade et al., 2020; Vanderlei et al., 2022), while regeneration heavily relies on resprouting (Barros et al., 2021; Marinho et al., 2016; Murphy et al., 1995; Noutcheu et al., 2023; Vanderlei et al., 2021). Despite providing some level of resilience to regenerating forests (Paz et al., 2018), the regenerative potential of resprouting is biased towards a few species that bear this strategy and may result in genetic impoverishment due to the absence of sexual reproduction (Noutcheu et al., 2023; Svensson et al., 2013). Although chronic disturbances can interact with environmental factors such as rainfall regime, soil fertility, and microclimate conditions, thereby intensifying severe abiotic conditions of dry forests (Silva et al., 2019), studies investigating their interactive effects on regeneration mechanisms are still limited (but see (Noutcheu et al., 2023). Furthermore, few studies have simultaneously assessed the relative contribution of different regeneration mechanisms and their underlying drivers and provided a comprehensive view of forest regeneration into human-modified landscapes, which is increasingly becoming the predominant scenario globally.
The Brazilian Caatinga is the world’s most extensive and diverse dry forest (da Silva et al., 2017). The Caatinga is an ecosystem with extreme climate conditions, including high temperatures, low and irregular rainfall, but with relatively fertile soils (da Silva et al., 2017). The human population living in the region is highly dependent on natural resources for their livelihood and impose a common set of disturbances/degradation such as slash-and-burn agriculture, wood extraction, exploitation of non-timber forest products, and cattle ranching, which have an enormous impact on the remaining forests ecosystems (da Silva et al., 2017; Ribeiro et al., 2015; Rito et al., 2017). Numerous studies have demonstrated that regeneration mechanisms in the Caatinga have been negatively affected by the slash-and-burn agriculture (Bezerra et al., 2023a, 2022), free-ranging livestock (goats and cattle) ranching (Lins et al., 2022), and leaf-cutting ant herbivory (Knoechelmann et al., 2020; Oliveira et al., 2023). Consequently, regenerating assemblages in the area are characterized by low density (i.e. low number of seedlings, sapling and resprouts) and impoverished diversity (de Paula et al., 2023; Vanderlei et al., 2022), with resprouting playing a crucial role in driving successional pathways and ensuring forest resilience (Barros et al., 2021; Noutcheu et al., 2023). In this context, Caatinga represents an interesting opportunity to investigate dry forest regeneration in a human-modified landscape.
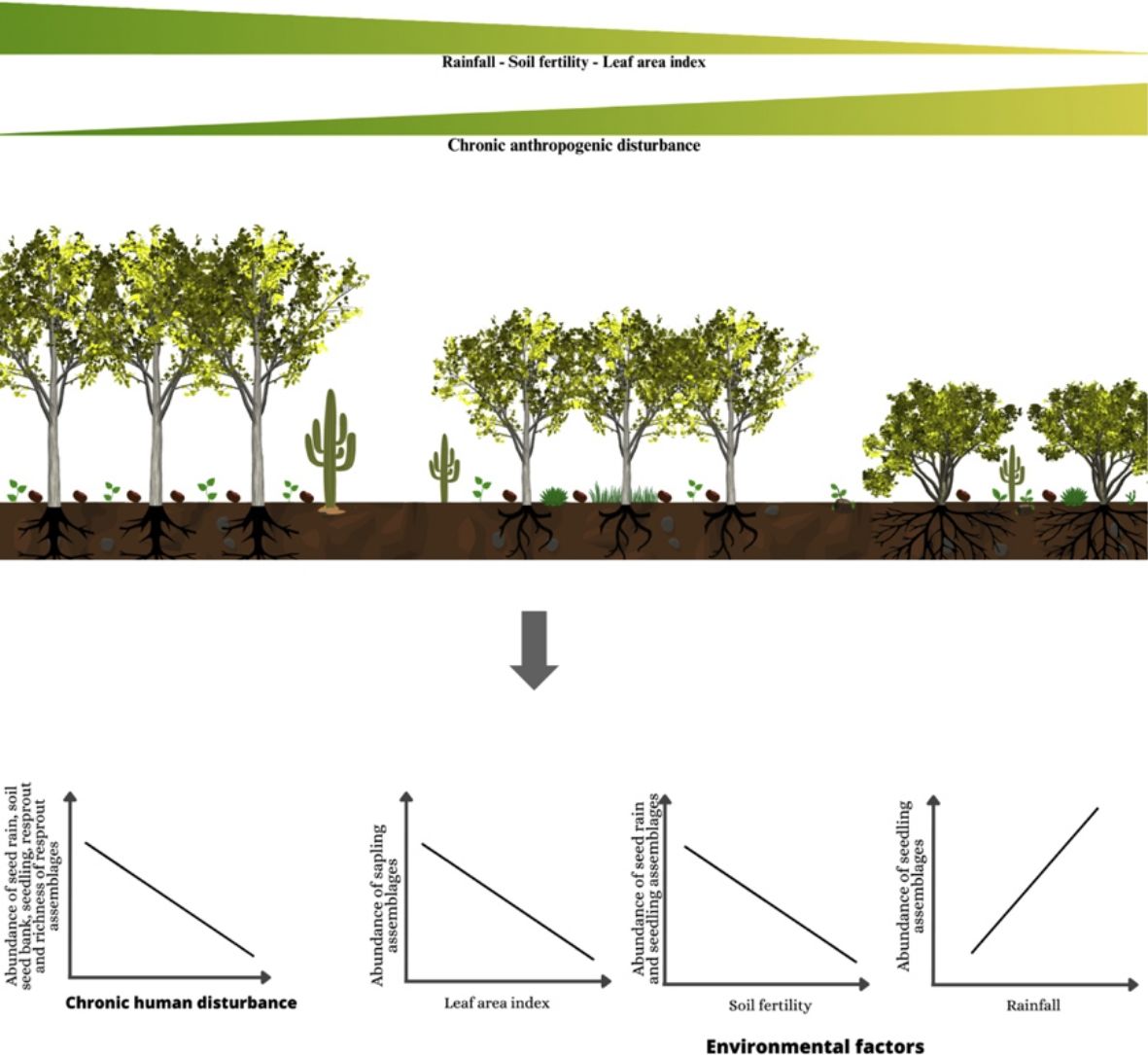
In this study, we evaluated the effects of chronic disturbance and environmental factors (rainfall, soil fertility, and light availability) on regeneration mechanisms (seed rain, soil seed bank, true seedling, true sapling, and resprout) in Caatinga dry forest. We classified seeds and woody plant regenerating assemblages as either autochthonous if they are present in the same plots with adults from the same species or allochthonous based on the absence of adults within of our study plots. Additionally, we assessed the adult-to-offspring ratio of the most widely distributed species in the focal landscape. Our general hypothesis is that disturbance and harsher environmental conditions will negatively affect reproductive regeneration mechanisms (seed rain, soil seed bank, true seedling, and true sapling) while promoting resprouting. We also hypothesize that, as disturbance and harsher environmental conditions increase, there will be a predominance of regenerants from autochthonous species and the adult-to-offspring ratio will decrease. Thus, we expected that increasing chronic disturbance, aridity, soil infertility (lower nutrient availably) and light availability will (1) negatively affect the abundance and richness of seed rain, soil seed bank, true seedlings, and true saplings while positively affecting resprouts, (2) rearrange the regenerating plant assemblages via the replacement of seeds and regenerants of disturbance-sensitive species by disturbance-adapted species, (3) increase the contribution of autochthonous species, and (4) decrease the adult-to-offspring ratio.
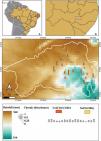
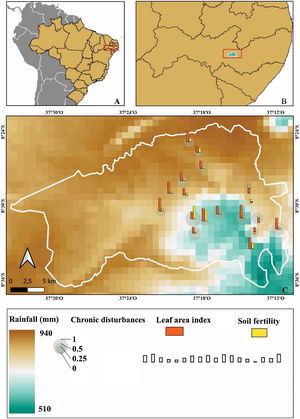
Materials and methodsStudy siteThe study was carried out in the Catimbau National Park (8°24'00″–8°36'35″ S; 37°00'30″–37°10'40″ W, Fig. 1), a protected area covering 607 km² in rural Pernambuco State, northeastern Brazil (Rito et al., 2017). The climate type is semi-arid bsh’ with the transition to the tropical rainy As’ according to the Köppen classification system (Alvares et al., 2013). The annual precipitation ranges between 440–1100 mm with a significant spatiotemporal variation (Rito et al., 2017). The vegetation is predominantly composed of low-stature dry forests, with the families Leguminosae, Euphorbiaceae, Myrtaceae, and Burseraceae being the most species-rich and dominant families (Rito et al., 2017).
Although the National Park was established in 2002, traditional farms (i.e., family smallholdings) still live within the area, and activities such as slash-and-burn agriculture, livestock farming (free-ranging goats), collection of living and dead wood, harvesting of non-timber forest products, and hunting are still common, leading to significant pressures on old growth forests (Rito et al., 2017).
Sampling designFieldwork was conducted between July 2018 and July 2020 in fifteen 0.1-ha (20 m × 50 m) plots for seed rain assessment and eighteen 0.1-ha plots for evaluating other ontogenetic stages. These permanent plots are part of the Catimbau ILTER project (https://www.peldcatimbau.org), which are distributed along independent gradients of chronic anthropogenic disturbance and annual rainfall (Rito et al., 2017; see also Fig. 1; more details about these gradients are presented below in the appropriate section.). All plots were situated on sandy soils with flat terrain and supported old-growth vegetation. The plots were separated by a minimum of 2 km and located within a total area of 21,430 ha (Rito et al., 2017).
Measurement of the chronic anthropogenic disturbances indexData on the level of chronic anthropogenic disturbance of each plot were obtained using the global multi-metric chronic disturbances index proposed by (Arnan et al., 2018). Concisely, this index considered the three most common sources of disturbance in the region: livestock pressure, wood extraction, and people pressure. These chronic disturbances indicators were measured using three distinct approaches: (1) Geographic distances based on remote sensing: using satellite imagery and ArcGIS 10.1 software, we measured the proximity of each plot to the nearest house and the nearest road. Since the level of disturbance is inversely related to distance, we used the inverse of distance as our metric; (2) Interviews with local inhabitants: the nearest village to each plot was identified using Geographical Information System and then informal and semi-structured interviews were conducted to assess the number of people in each village, which was weighted by distance from the plots; and, (3) Measures of disturbance in the field: within each plot, we conducted field assessments to measure the length of goat trails, the extraction of alive wood (stem cuts), and the extraction of coarse woody debris (litter). The values of these indicators of disturbance (i.e. proximity to the nearest house, proximity to the nearest road, number of people in each village, goat trail length, alive wood extraction, and coarse woody debris extraction) were standardized between 0 and 1, which makes disturbance metrics of different units comparable and easily to combine in the same index. The index ranged from 2 to 58 (from the lowest to the highest disturbance intensity) among the plots. For more details on the calculation of the disturbance index, please see Arnan et al. (2018).
Environmental variablesBased on previous studies (Rito et al., 2017; Silva et al., 2019), mean annual rainfall data were extracted from the WorldClim global climate repository (Fick and Hijmans, 2017). The closest climate stations to the study sites were utilized to obtain these rainfall data, with a spatial resolution of 1 km, using the maptolls package in R 3.6.1 (Bivand and Lewin-Koh, 2020). This package enabled the calculation of the mean annual rainfall for each plot, which ranged from 510 mm to 940 mm.
As suggested by (Sfair et al., 2018), soil fertility for each plot was assessed by randomly selecting three soil samples measuring 10 cm × 10 cm from a depth of 0–30 cm. The soil samples were then sent for laboratory analysis of soil physicochemical properties related to nutrient availability (Al, H, S, P, CTC, Ca, pH, K), water content, organic matter, and acidity. These analyses followed the procedures recommended by the Brazilian Ministry of Agriculture for soil analyses (EMBRAPA, 1997). The soil fertility of each plot was determined using the formula SF = [Ca + Mg + K − log(1 + A1)] × OM + 5 (Lu et al., 2002), where SF = soil fertility, Ca = exchangeable calcium (cmolc dm−3), Mg = exchangeable magnesium (cmolc dm−3), K = exchangeable potassium (cmolc dm−3), Al = exchangeable aluminium (cmolc dm−3), and OM = organic matter (g kg−1). The soil fertility values ranged from 10.8–59.6 among the plots. More details on soil sampling and physical/chemical analyses can be found in Sfair et al. (2018).
To assess light availability in each plot, we estimated the leaf area index throughout 10 hemispherical photographs taken at a distance of 1.50 m from the ground level, at intervals of 5 meters, and either at dawn before sunrise or at dusk after sunset to prevent direct solar radiation in any part of the canopy (Whitmore et al., 1993). Photographs were taken again across the same plots and at the same time three times during the year 2016 within the seasons. We analyzed the hemispherical photographs with the Gap Light Analyser software (GLA, version 2.0) in order to obtain the leaf area index of each plot (Frazer et al., 1999), which ranged from 0.02 to 0.48.
Sampling of regeneration mechanismsSeed rain – Seed traps composed of a deep squared plastic tray with an effective trapping area were used to collect the seed rain. Holes were drilled through the tray to drain out rainwater. We randomly placed 5 seed traps of 1 m × 1 m at 1 m of the soil level in each plot. Seed rain was collected monthly between July 2018 and July 2020. The materials in the cloths were carefully screened and identified taxonomically in the laboratory. All visible and healthy seeds were counted and used for the analyses.
Soil seed bank – Forty soil samples were randomly collected in 20 cm × 20 cm × 5 cm squares in each plot, ten soil samples at the end of both dry and rainy seasons for two consecutive years (from July 2018 to July 2020). To assess seed viability, a tetrazolium test was conducted. Seeds were immersed in a 1% aqueous solution of 1% triphenyl tetrazolium chloride (TTC), then they were placed in Petri dishes with distilled water, covered with aluminum foil, and stored at a temperature of 35 °C for four hours. After the incubation, the seeds were washed in running water for observation of the embryo. Seeds were classified as viable if a red spot appeared on the seed tissue (indicating positive seed viability) or non-viable if no spot formation occurred (indicating negative seed viability) (França-Neto and Krzyzanowski, 2019).
Regenerating assemblages – In each plot, seedlings, saplings, and resprouts of woody species were sampled using standard forestry methods (Pancel and Köhl, 2016). Seedlings were defined as individuals with a height <1 m, while saplings were individuals with a height between 1 and 1.5 m and a diameter at soil base <3 cm (Ribeiro et al., 2015). Seedling communities were sampled in nine 2 m × 1 m subplots, and saplings in three 5 m × 5 m subplots, both located in the center of each 50 m × 20 m plot and separated by 10 m. All plants were identified at the species level according to the botanical nomenclature followed by the Angiosperms Phylogeny Group (APG IV, 2016) and Brazilian flora list. To determine if the seedlings and saplings were resprouts, a 30 cm hole was dug around each stem as needed. Individuals were classified as true seedlings/saplings if they were not connected to an adult tree, or as resprouts if they were connected to an adult individual that was not a resprout or a ramet according to Noutcheu et al. (2023).
Adult assemblages – In order to classify seeds and woody plant regenerating assemblages as either autochthonous or allochthonous based on the presence of adult individuals within the plot, we used data on adult assemblages from (Rito et al., 2017), who surveyed the permanent plots (i.e., the 18 50 m × 20 m plots) of the Catimbau ILTER project. Adults were defined as individuals with a diameter at soil level >3 cm and a height >1.5 m (Rito et al., 2017). Additionally, we used the adult assemblages to determine the natural regeneration capacity of the species based on the adult-to-offspring ratio (Pausas and Keeley, 2014).
Statistical analysesTo determine the spatial independence of plots, the species composition similarity among plots (Bray-Curtis index), and the inter-plots distance matrix (one per each regeneration mechanism), we ran a Mantel test. Mantel results indicated spatial independence among our plots for all regeneration mechanisms: seed rain (r = 0.104, p = 0.16), soil seed bank (r = 0.09, p = 0.115), woody plant regenerating assemblages (r = 0.07, p = 0.201), and adult (r = 0.05, p = 0.38).
In order to assess whether the species composition across regeneration mechanisms was recorded adequately, given that the sampling area of each regeneration mechanism was different, the sample coverage was performed (Chao and Jost, 2012) using the following equation:
where f1 and f2 represent the number of species with one and two individuals respectively, and n is the number of individuals. The mean ± standard deviation of the sample coverage was > 0.9% for each regeneration mechanism (seed rain 0.98 ± 0.03; soil seed bank 0.94 ± 0.02; woody plant regenerating assemblages 0.91 ± 0.03 and adults 0.99 ± 0.01), indicating adequate sampling coverage. To examine the taxonomic dissimilarity among the four ontogenetic stages, we calculated the Bray-Curtis dissimilarity matrix for the ontogenetic stages and compared them using the Mantel test.
To determine the structure of all regeneration mechanism assemblages in each plot, we calculated the density, and species richness of the seed rain, soil seed bank, seedlings, saplings, resprouts, and adults. Additionally, the status, sources of seed dispersal and the uses of some species (more abundant) were also informed. To compare species richness among regeneration mechanisms, we used the interpolation and extrapolation methods in the iNEXT package (Chao et al., 2014). Due to discrepancies in terms of abundance and species richness, we focused the statistical comparisons on the woody plant regenerating assemblages (seedlings, saplings, and resprouts), using Generalized Linear Models (GLM) followed by pairwise contrast analyses. Additionally, we used the adult-to-offspring ratio (Pausas and Keeley, 2014) to determine the natural regeneration capacity of the most abundant adult species.
To examine the isolated and interactive influences of the explanatory variables on the abundance and species richness of each regeneration mechanism (i.e., seed rain, soil seed bank, seedlings, saplings, and resprouts) on the adult-to-offspring ratio, and the number of allochthonous regeneration mechanisms, we used a model selection approach. We built generalized linear models (GLM) for the woody plant regenerating assemblages and adult-to-offspring ratio and generalized linear mixed models (GLMM) for the seed rain and soil seed bank. For all models, except the adult-to-offspring ratio, we used a Poisson distribution error and, in the case of overdispersion, a quasi-Poisson distribution error was applied. For the adult-to-offspring ratio, we used a binomial distribution (link = logit). Candidate models for each response variable (species richness and abundance) were chosen based on our explanatory variables and empirical support. Then, Akaike’s information criterion with a correction for small samples (AICc) was used to select the best models. In the case of overdispersion, qAICc was used instead of AICc to rank the models (Calcagno and Mazancourt, 2010). To select the best models, set models with AICc (or qAICc) differences lower than 2 were chosen (Burnham and Anderson, 2002). The best-supported models, which included important predictors beyond the intercept, were identified. The dredge function in the MuMin package in R was used for model selection (Δ ((Q)AICc) < 2). To assess the collinearity among the predictor variables, we calculated the variance inflation factor (VIF) of each predictor using the car package for R (version 3.0.1) (R, 2016). All VIF values were lower than 1.46, with soil fertility = 1.26, rainfall = 1.45, chronic disturbances = 1.24, and leaf area index = 1.11, indicating independence among the predictors (Jou et al., 2014) and considered suitable for further analyses.
The effects of chronic disturbance and environmental conditions on the species composition of each regeneration mechanism were assessed using non-metric multidimensional scaling (NMDS). Then, the envfit method from the vegan package was used to obtain the effect of each variable on the composition of each regeneration mechanism (Oksanen et al., 2019). Data that did not meet the homoscedasticity criteria were log(x) +1 transformed. Pairwise contrast analyses were conducted using the contrast function from the emmeans package (version 1.8.4−1); (Lenth, 2023; Searle et al., 1980)). The GLMM models were built using the lme4 package (version 1.1–7) (Bates et al., 2015), with plots as the random factor. In the case of overdispersion, the bblme package (version 1.0.23.1) (Bolker and R Development Core Team, 2020) was used to build the models. Additionally, a marginal R2 for each selected model was calculated with the method proposed by (Nakagawa and Schielzeth, 2013). All analyses were performed in R (R, 2016).
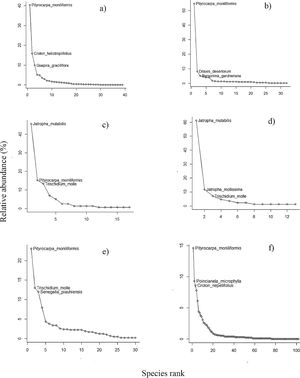
ResultsTaxonomic attributes of regenerating and adult tree assemblagesOver the course of two years, a total of 5,239 seeds were collected in the seed rain, along with 932 in the soil bank, representing 40 and 32 species, and 15 and 12 families, respectively (Table 1). Shannon diversity index ranged from 4.4 in saplings to 27.9 in adults (Table 1). Overall, the assemblages of all forest mechanisms were predominantly dominated by a small set of species (3–5), which were both frequent and accounted for most of the individuals (Table S1, Supplementary material) (Fig. 2a and e). In the seed rain, Pityrocapa moniliformis, Cnidoscolus bahianus, and Senegalia piauhiensis were the most widely distributed species, occurring in 12, 10, and 10 plots, respectively (Table S1, Supplementary material). In terms of abundance, Pityrocarpa moniliformis, Croton heliotropiifolius, and Guapira graciliflora accounted for over 66% of all seeds, while the 25 least abundant species had no more than 50 seeds each (Fig. 2). Regarding the soil seed bank, Pityrocapa moliliformis, Byrsonima gardneriana, and Commiphora leptophloeos were the most frequent species, occurring in 10, 9 and 9 plots, respectively (Table S1, Supplementary material). Pityrocapa moliliformis and Ditaxis desertorum accounted for more than 59% of all seeds, while the remaining 30 species had less than 50 seeds (Fig. 2). Among seedlings, Jatropha mutabilis and Pityrocapa moliliformis were the most widely distributed species, occurring in 11 and 4, plots, respectively (Table S1, Supplementary material). Three species alone accounted for 73.4% of all individuals (Jatropha mutabilis, Pityrocapa moliliformis, and Trischidium molle), while the 12 least abundant species (representing 70% of all species) had no more than 5 individuals each (Fig. 2). In the sapling assemblages, the genus Jatropha represented 83.5% of 85 individuals collected across all studied plots, and among the thirteen species, only three species had more than 5 individual each (Fig. 2). Jatropha mutabilis and Jatropha mollissima were the most distributed species, occurring in 10 and 4 plots, respectively (Table S1, Supplementary material). Among resprouts, Trischidium molle, Pityrocapa moliliformis, and Poincianela mycrophyla were the most frequently occurring species, found in 11, 9, and 8 plots, respectively (Table S1, Supplementary material). Pityrocapa moliliformis, Trischidium molle, and Senegalia piauhiensis accounted for more than 48% of all resprouting individuals, while the remaining 27 species had less than 35 individuals (Fig. 2). Additionally, a substantial proportion of species across the assemblages were rare in terms of both frequency and total abundance. Finally, the assemblages shared a small number of species on average (3.3%), particularly those considered dominant.
Community characteristics of regeneration mechanisms and trees adults in 18 0.1-ha plots surveyed in the Caatinga dry forest, Catimbau National Park, Northeast Brazil.
| Characteristics of sample and community | Seed rain | Seed soil | Seedlings | Saplings | Resprouts | Adults |
|---|---|---|---|---|---|---|
| Plots | 15 | 18 | 162 | 54 | 162 | 18 |
| Abundance (total nº of individual) | 5239 | 932 | 158 | 85 | 416 | 3896 |
| Observed species (nº) | 39 | 32 | 17 | 13 | 30 | 97 |
| Estimated richness (mean + SE) | 43.49 ± 8.31 | 49.9 ± 23.5 | 21.47 ± 10.05 | 21.89 ± 9.37 | 36.23 ± 8.85 | 100 ± 2.80 |
| Shannon diversity | 9.01 | 7.26 | 6.35 | 4.44 | 15.49 | 27.91 |
| Estimated Shannon diversity (mean + SE) | 9.05 ± 0.18 | 7.44 ± 0.36 | 6.83 ± 0.74 | 5.07 ± 0.94 | 16.21 ± 0.91 | 28.29 ± 0.57 |
| Species with one or two individuals (%) | 10 | 7 | 9 | 8 | 7 | 23 |
Species accumulation curves for the three size classes surveyed in this study: (a) seed rain, (b) soil seed bank, (c) true seedling, (d) true sapling, (e) resprout, and (f) adult, as functions of the number of individuals sampled in the 18 0.1-ha m2 plots in the Caatinga dry forest, Catimbau National Park, Northeast Brazil.
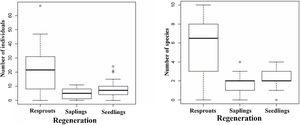
During the 25-month monitoring period, the accumulated seed rain ranged from 0.2 to 423 seeds per m2 across the stands (Table S2, Supplementary material), with an average species richness of 9.2 ± 0.73 species per m2 per stand (average ± SD). The soil seed bank exhibited variation in density, ranging from 0.07 to 35.5 seeds/m2, with an average species richness of 6.61 ± 0.61 species per m2 (Table S2, Supplementary material). Considering the woody plant regenerating assemblages, seedling density varied from 0 to 5.99 individuals per m2 across forest stands, while saplings varied from 0 to 0.69 individuals per m2, and resprout density varied from 0 to 1.28 individuals per m2 (Table S2, Supplementary material). Species richness exhibited a similar trend, ranging from 1.61 to 5.22 among these three regenerating mechanisms (seedling, sapling, and resprout). Despite such a low density and reduced species richness, these regenerating mechanisms differed significantly in terms of abundance and species richness attributes (Fig. 3. F = 37.72, df = 2, P < 0.001; F = 46.96, df = 2, P < 0.001, respectively). Resprouts supported the highest scores, followed by seedlings (Fig. 3); i.e., 21.6 % more resprouts as compared to seedlings and saplings. Resprouting from stems was the most frequent resprouting type (F = 220.61, df = 2; P < 0.001, Fig. S1 Supplementary material). In woody plant regenerating assemblages, resprouts accounted for 63% of all individuals and, among the most abundant species, the genus Jatropha was the only that did not exhibit any resprouts.
Regenerating assemblages and adultsRegenerating assemblages did not correlate taxonomically with the adult assemblages, as follows: seed rain (r = 0.66, p = 0.001), soil seed bank (r = 0.52, p = 0.001), and regenerating assemblages (r = 0.55, p = 0.001). Accordingly, a substantial proportion of the species attending the regenerating assemblages was classified as allochthonous: 52% of seed rain, 50% of the soil seed bank, 22% of seedling, 31% of sapling, and 33% of resprout. The regeneration capacity, as measured by the adult-to-offspring ratio, varied among the most abundant species and correlated with the regeneration mechanisms, with seedlings achieving the highest scores (0.01−0.27), followed by resprouts (0−0.15), and saplings (0−0.07) (Table 2).
Tree species, status, seed dispersal type, types of use, and adult-to-offspring ratio surveyed in 18 0.1-ha plots in the Caatinga dry forest, Catimbau National Park, Northeast Brazil. Types of use are: Me: medicinal, Ct: construction, Or: ornamental, Fd: food, Fl: fuel, Ot: other.
| Species | Status | Seed dispersal | Uses | Seedling/adult | Sapling/adult | Resprout/adult |
|---|---|---|---|---|---|---|
| Pityrocarpa moniliformis | Native | Autochory | Me, Ct, Ot | 0.03 | – | 0.15 |
| Poincianela microphylla | Native | Autochory | Me, Ct, Ot | 0.02 | – | 0.08 |
| Croton nepetifolius | Native | Autochory | Me, Fl | 0.01 | 0.005 | 0.01 |
| Croton heliotropiifolius | Native | Autochory | Me, Fl | 0.01 | 0.03 | 0.06 |
| Jatropha mutabilis | Native | Autochory | Me | 0.27 | 0.07 | – |
Overall, all regeneration mechanisms responded to the analyzed drivers, although responses were specific, and a significant proportion of the variation remained unexplained.
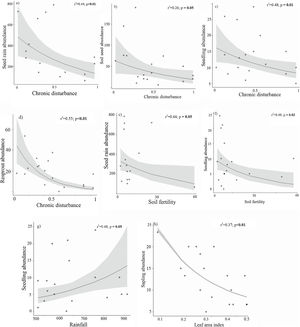
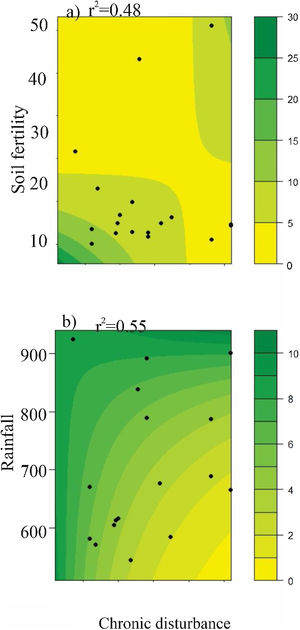
AbundanceBriefly, the abundance of seeds (both seed rain and seed bank), seedlings, and resprouts were negatively affected by chronic disturbances (Table 3; Fig. 4a–d, respectively). Additionally, both seed rain and seedling abundances were also negatively affected by soil fertility (Table 3; Fig. 4e and f, respectively), while seedling abundance responded positively to rainfall (Table 3; Fig. 4g). Sapling abundance was negatively affected by the leaf area index across forest stands (Table 3; Fig. 4h). Furthermore, certain environmental factors had contingent effects depending on the level of chronic disturbance. For example, the negative impacts of soil fertility on seedling abundance and rainfall on resprout abundance were greater in more disturbed areas (Table 3; Fig. 5a and b, respectively).
Results from generalized linear (GLM) and mixed models (GLMM) of the abundance and richness of seed rain, soil seed bank, true seedling, true sapling, resprouts, and woody plant regenerating assemblages in response to chronic anthropogenic disturbances and abiotic factors in 18 0.1-ha plots surveyed in the Caatinga dry forest, Catimbau National Park, Northeast Brazil.
| Response variables | Explicative variables selected | d.f. | t | P-value |
|---|---|---|---|---|
| Seed rain abundance | Chronic disturbance | 6 | −2.908 | <0.01 |
| Rainfall | 6 | 1.499 | 0.11 | |
| Soil fertility | 6 | −1.955 | 0.05 | |
| Leaf area index | 6 | 0.934 | 0.3 | |
| Soil seed bank abundance | Chronic disturbance | 4 | −1.912 | 0.05 |
| True seedling abundance | Rainfall | 6 | 2.087 | 0.05 |
| Chronic disturbance | 6 | −2.893 | 0.01 | |
| Soil fertility | 6 | −2.525 | 0.02 | |
| Leaf area index | 6 | −1.112 | 0.28 | |
| Chronic disturbance * soil fertility | 6 | 2.53 | 0.02 | |
| True sapling abundance | Leaf area index | 2 | −3.310 | <0.01 |
| Resprout abundance | Rainfall | 4 | −1.464 | 0.165 |
| Chronic disturbance | 4 | −3.254 | <0.01 | |
| Chronic disturbance * rainfall | 4 | 2.810 | 0.01 | |
| Seed rain richness | Leaf area index | 3 | 1.919 | 0.05 |
| Soil seed bank richness | leaf area index | 3 | 1.348 | 0.17 |
| True seedling richness | Chronic disturbance | 2 | −1.543 | 0.12 |
| True sapling richness | Leaf area index | 2 | −0.75 | 0.42 |
| Resprout richness | Rainfall | 4 | −0.567 | 0.57 |
| Chronic disturbance | 4 | −3.032 | <0.01 | |
| Chronic disturbance * rainfall | 4 | 2.757 | <0.01 | |
| Seed rain allochthonous | Chronic disturbance | 2 | −0.889 | 0.374 |
| Soil seed bank allochthonous | Soil fertility | 2 | −0.801 | 0.423 |
| Seedling allochthonous | Rainfall | 2 | 2.828 | <0.01 |
| Sapling allochthonous | Rainfall | 2 | 0.912 | 0.362 |
| Resprout allochthonous | Rainfall | 2 | 2.383 | 0.01 |
| Seedling/adult ratio | Rainfall | 3 | 1.511 | 0.131 |
| Soil fertility | 3 | −0.978 | 0.328 | |
| Sapling/adult ratio | Rainfall | 3 | 1.483 | 0.138 |
| Soil fertility | 3 | −9.959 | 0.338 | |
| Resprout/adult ratio | Rainfall | 2 | 0.708 | 0.479 |
Relationships between abundance of seed rain, soil seed bank, true seedling, true sapling and resprout with chronic disturbance, soil fertility, rainfall, and leaf area index in the Caatinga dry forest, Catimbau National Park, Northeast Brazil. Models were developed using log transformed data for some species.
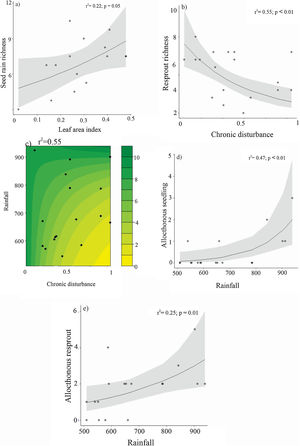
Species richness exhibited less responsiveness, with seed rain positively affected by leaf area index, while the resprout assemblage responded negatively to chronic disturbance (Table 3; Fig. 6a and b, respectively). Resprout species richness also demonstrated interactive effects between chronic disturbances and rainfall (Table 3; Fig. 6c), with rainfall having a greater negative impact in more disturbed areas. The presence of allochthonous species in both seedlings and resprouts was positively related to rainfall, and neither chronic disturbance nor environmental factors were associated with theadult-to-offspring ratio (Table 3; Fig. 6d and e).
Relationships between richness of seed rain (a), resprout (b), woody plant regenerating assemblages (c), and leaf area index and chronic disturbance, and combined effect of chronic disturbance and rainfall on (d) resprout and (e) regenerating assemblages richness in the Caatinga dry forest, Catimbau National Park, Northeast Brazil.
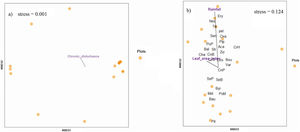
Finally, the species composition across the regeneration mechanisms was influenced by the analyzed drivers as follows: sapling assemblages strongly responded to chronic disturbance (Fig. 7a; R2 = 0.39, P = 0.01), while resprouts responded to rainfall (Fig. 7b; R2 = 0.61, P < 0.01) and leaf area index (Fig. 7b; R2 = 0.35, P = 0.04).
Non-metric multidimensional scaling (NMDS) showing the association of environmental factors with species composition of (a) true sapling and (b) resprouts surveyed in the 18 0.1-ha m2 plots in the Caatinga dry forest, Catimbau National Park, Northeast Brazil. The acronyms stand for Aca = Acalypha brasiliensis, Bal = Balfourodendron molle, Bau = Bauhinia acuruana, Bou = Bouxera sp, Byr = Byrsonima gardneriana, Cha = Chamaecrista zygophylloides, CnB = Cnidoscoleus bahianus, CnP = Cnidoscoleus pubescens, com = Commiphora leptophloeos, Cos = Cospidaria agentia, CrH = Croton heliotropiifolius, CrN = Croton nepetifolius, Ery = Erythroxylum revolutum, Eug = Eugenia sp2, Mel = Melochia tomentosa, Neo = Neocalyptrocalyx longifolium, Oxa = Oxalis sp, pel = Peltogyne pauciflora, Pip = Piptadenia stipulacea, Pit = Pityrocarpa moniliformis, PoM = Poincianella microphylla, PoP = Poincianella pyramidalis, SeB = Senegalia bahiensis, SeP = Senegalia piauhiensis, Sen = Senna acuruensis, Sti = Stigmaphyllon paralias, Str = Strychnos rubiginesa, Tri = Trischidium molle, Var = Varonia leucoxyflora, Ziz = Ziziphus joazeiro.
Our results suggest that the Caatinga dry-forest regeneration relies on low-density and taxonomically impoverished seed rain, soil seed bank, and woody-plant regenerating assemblages (seedlings, saplings, and resprouts). The combined density of saplings and resprouts averaged 0.565 individuals per m2. However, different forest regeneration mechanisms play contrasting roles, with resprouts, particularly from stumps, being more prevalent compared to seedlings and saplings. In fact, Caatinga forests on sandy soils appear to support more than twice as many resprouts than seedlings and saplings. As expected, Caatinga regeneration mechanisms respond to a combination of drivers that pose different effects on distinct assemblage attributes. While chronic disturbances shape the structure of seed rain, soil seed bank, and seedling assemblages, aridity alters species composition across resprouting assemblages. Moreover, regeneration drivers pose interactive effects, making the process of Caatinga forest regeneration complex, with chronic disturbances playing a prominent role. The limited similarity between regenerating and adult assemblages suggests that a large number of species have limited capacity to achieve the adult phase and thus effectively contribute to the Caatinga dry forest regeneration.
There is still little information about regeneration mechanisms (seeds, seedlings, saplings, and resprouts) across dry forests. Consequently, this makes generalization difficult. In general, our findings are in accordance with previous studies about the low numbers of seeds and woody plant assemblages in tropical dry forests around the world (Kennard et al., 2002; Khurana and Singh, 2001; Murphy et al., 1995; Murphy and Lugo, 1986; Sampaio et al., 2007; Skoglund Jerry, 1992; Swaine, 1992), including the Caatinga dry forest (Barros et al., 2021; Ribeiro et al., 2015; Vanderlei et al., 2022, 2021). Regardless of the abundance and density of regeneration mechanisms, disturbances and environmental factors have been consistently reported to negatively impact these mechanisms. For instance, studies have demonstrated that seed rain and soil seed bank were negatively associated with disturbances in dry forests (Bezerra et al., 2023a, 2022; Kennard et al., 2002). (Trindade et al., 2020) reported that water availability played a more significant role than time since abandonment (4–70 years) in shaping a functionally structured seedling assemblage in a chronosequence of forest regeneration. Moreover, (Barros et al., 2021; Vanderlei et al., 2021) suggested that the regeneration of areas that previously supported slash-and-burn agriculture mostly relies on resprouting. However, disturbances may also have a negative effect even on resprouts, as demonstrated in dry forests (Kennard et al., 2002), deciduous forests (Ferreira et al., 2017), and savanas (Franklin et al., 2010). The lack of similarity between regenerating and adult assemblages has also been found across dry forests (Hopfensperger, 2007; Lemenih and Teketay, 2006). Although resprouting has been documented as an important regeneration mechanism in dry forests (Ferreira et al., 2017; Jaureguiberry et al., 2020). Kennard et al. (2002) have found that seedlings were more important in terms of abundance and density in lowland Bolivian dry forests. In contrast, resprouting from roots has been found as the most frequent mechanism of regeneration in dry forests of Central Brazil (Scariot et al., 2008).
The sparse and impoverished seed rain, seed soil bank, and regenerating assemblages are likely to result from a combination of processes, including (1) low production of viable seeds, which is evident in both the seed rain (12% of viable seed in Bezerra et al., 2023b) and the soil seed bank (i.e., 5–164 seeds m² (Lins et al., 2022; de Paula et al., 2023) ); (2) high mortality of seeds due to fire from human activities, such as slash-and-burn agriculture (i.e., seeds were 15 times lower in burned plots compared to control plots; Bezerra et al., 2023a), as Caatinga seeds are not adapted to fire like those in fire-prone ecosystems (Bezerra et al., 2022); and (3) reduction of seedlings survival by goats (i.e., seedlings were 29.4 times more abundant in goats exclosure plots than in plots with free access; Lins et al., 2022) and leaf-cutting ants (i.e., control plots had 60% and 44% more seedlings than ant nest and foraging areas, respectively; Knoechelmann et al., 2020). The free-ranging goats observed in Caatinga are generalists and consume all plant-related items, leading to intensive browsing of seedlings (Lins et al., 2022; Menezes et al., 2021). Furthermore, the proliferation of leaf-cutting ants due to anthropogenic disturbances (Siqueira et al., 2017) leads to intensified leaf consumption and herbivory rates in the Caatinga (Siqueira et al., 2018), significantly impacting seeds (Oliveira et al., 2023), and seedlings and saplings (Knoechelmann et al., 2020), ultimately affecting natural regeneration in the area. The challenging conditions of the Caatinga, characterized by open and desiccated landscapes with high temperatures and limited water availability, impose significant environmental filters that impede regeneration. In addition to ecological filtering, the composition of regenerating assemblages may be explained by dispersal limitations caused by the removal of seed sources due to anthropogenic disturbances, such as slash-and-burn agriculture (Bezerra et al., 2023a). Moreover, the presence of highly competitive herbaceous species, such as some Malvaceae species in our focal landscape (Vieira et al., 2022), can also affect seed germination and seedling establishment due to resource competition (Holz et al., 2009). Given the greater abundance of herbaceous species in the Caatinga vegetation compared to juvenile woody species (Knoechelmann et al., 2020; Menezes et al., 2021), their role as barriers to the recruitment by trees can also explain some of our results.
The higher frequency of resprouting from stems can be explained by the coppicing activities practiced by local residents who use certain parts of the plant for their needs (Table 3). However, some species can lose the resprouting ability with repeated disturbances (Bellingham and Sparrow, 2000), which may explain the highest abundance and species richness of resprouts found in the least disturbed plots (Table 2). Additionally, bud banks in stems, responsible for resprouting, are highly sensitive to disturbances in dry tropical forests (Ferreira et al., 2017). Although tropical forests regenerate primarily via resprouting following disturbances (Chazdon, 2017; Vieira and Proctor, 2007), the reasons why resprouting is a particularly relevant regeneration mechanism in the Caatinga are not fully understood, as multiple viewpoints exist. The first one suggests that the ability to resprout depends on the rainfall regime, frequency, intensity, and duration of disturbance (McDonald et al., 2010). Another viewpoint proposes that the resprouting ability results from ancestral conditions, and the failure of resprouting is an evolutionary-derived trait (Clarke et al., 2013). Moreover, according to Pausas and Keeley (2014), resprouts receive resources from adult individuals, whereas true seedlings do not, giving them advantages in low-resource environments such as disturbed areas with desiccating climatic regimes and exhausted soils. However, it is worth noting that Jatropha mutabilis, which is one of the most abundant species among adults, true seedlings, and true saplings, apparently does not regenerate via resprouting, which can be related to the lack of carbohydrates reserves (Vanderlei et al., 2021). Given these contrasting trends, there is clearly much more to be learned about resprouting in the Caatinga.
Rainfall emerges as an important driver of the resprout composition. Our results also indicate an interaction between rainfall and disturbance, as the reduced rainfall had a more pronounced impact on disturbed areas (Figs. 6 and 7). It is well-established that rainfall represents a strong environmental factor in dry forest germination, establishment, and survival of plant species (Ceccon et al., 2006; Chazdon et al., 2007). In fact, we observed that aridity and leaf area index were the only environmental factors related to the species composition of resprouts and true saplings, albeit not with abundance and richness. This indicates the operation of compensatory dynamics within the communities, where some species thrive while others decline in response to reduced rainfall. In this context, species possessing traits such as resprouting ability or germination limited to rainy periods may exhibit greater tolerance and consequently increase their dominance (Ceccon et al., 2006; Lieberman and Li, 1992). The contrasting effect of light on regeneration mechanisms is probably related to the frequent and constant disturbances in the plots. The canopy structure and sparse tree cover of the Caatinga allow light penetration through the canopy to the ground, yet regeneration of initial growth stages requires shade (Ricardo et al., 2008) and moisture (Ceccon et al., 2006). On the other hand, the negative effect of soil fertility may suggest that the increase in dominance is not directly linked to enhanced soil fertility.
In summary, our study revealed the negative effects of chronic disturbance on seed rain, soil seed bank, true seedlings, true saplings, and resprouts in the Caatinga vegetation. These drivers affected mostly true seedling and sapling abundance, as well as the persistence of resprouts, while aridity altered species composition. Given that (1) small-size individuals are more susceptible to these disturbances and harsh environmental conditions, (2) the projected increase in chronic disturbances due to the presence of the local population who still live within the park (Albuquerque and Melo, 2018), and (3) rainfall is predicted to decrease in the region (Magrin et al., 2014), we expect even stronger impacts on regeneration mechanisms in the near future. Thus, measures should be taken in order to limit the ongoing drivers in the area, which can lead to desertification if nothing is done to safeguard the Caatinga vegetation.
Declaration of competing interestThe authors declare that they have no financial or non-financial conflicts of interest.
We thank Catimbau National Park landowners for giving us permission to work on their proprieties. We are grateful to Dani, Lays, and David for fieldwork assistance. This study is part of the PhD thesis of R. Noutcheu supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq processes 153811/2017-0) at the Universidade Federal de Pernambuco. F.M.P. Oliveira thanks CAPES and FACEPE for her postdoctoral grants (processes 88887.163451/2018-00 and BCT-0073-2.05/18), M. Tabarelli and I.R. Leal also thank CNPq for productivity grants (processes 310228/2016-6 and 305611/2014-3, respectively). This study was supported by the CNPq (PELD process 403770/2012-2 and Edital Universal process 470480/2013-0), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES process 001), Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE process APQ-0738-2.05/12). Laura Snook and Carine Emer provided helpful comments on an earlier version of this paper.