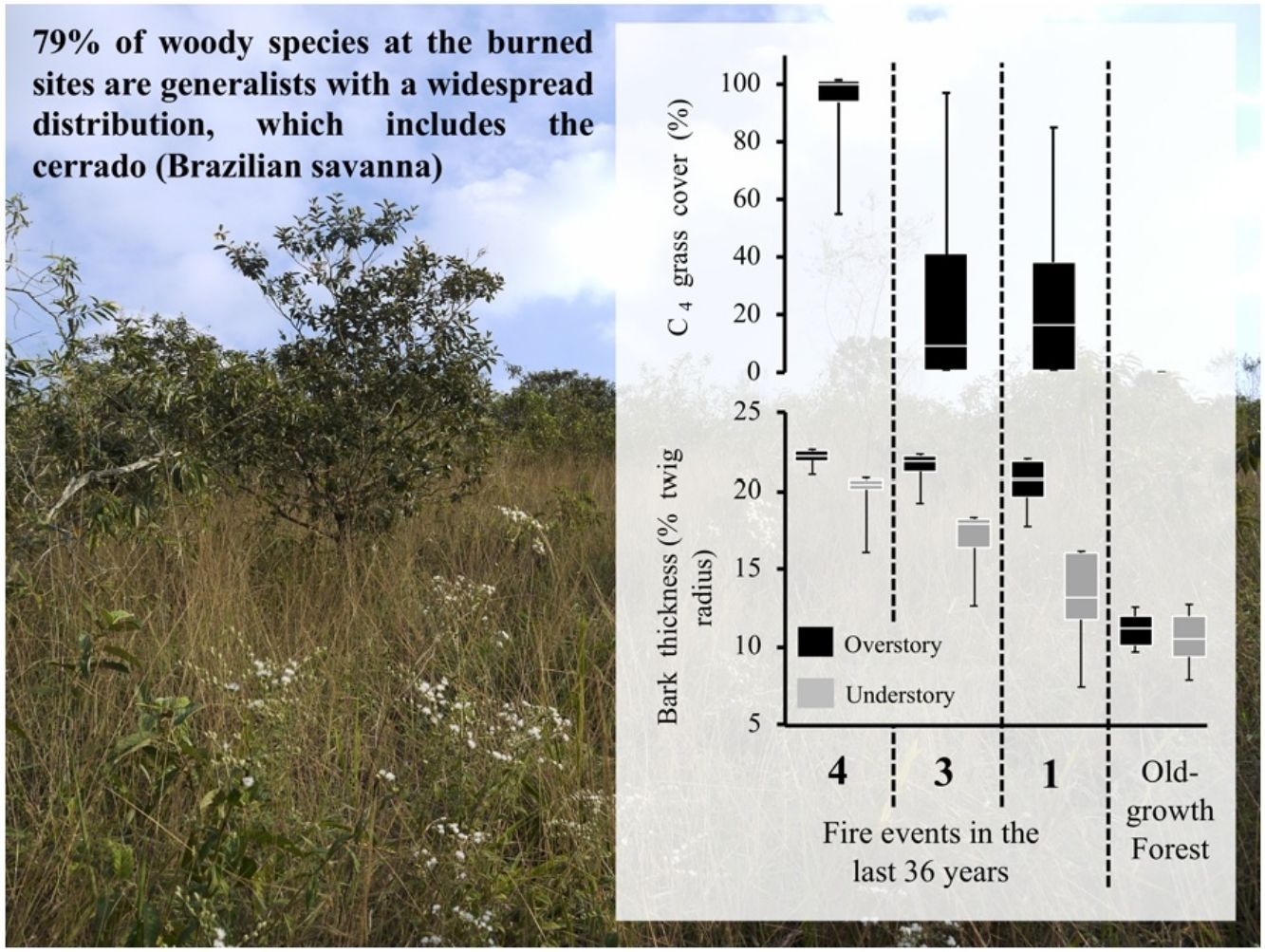
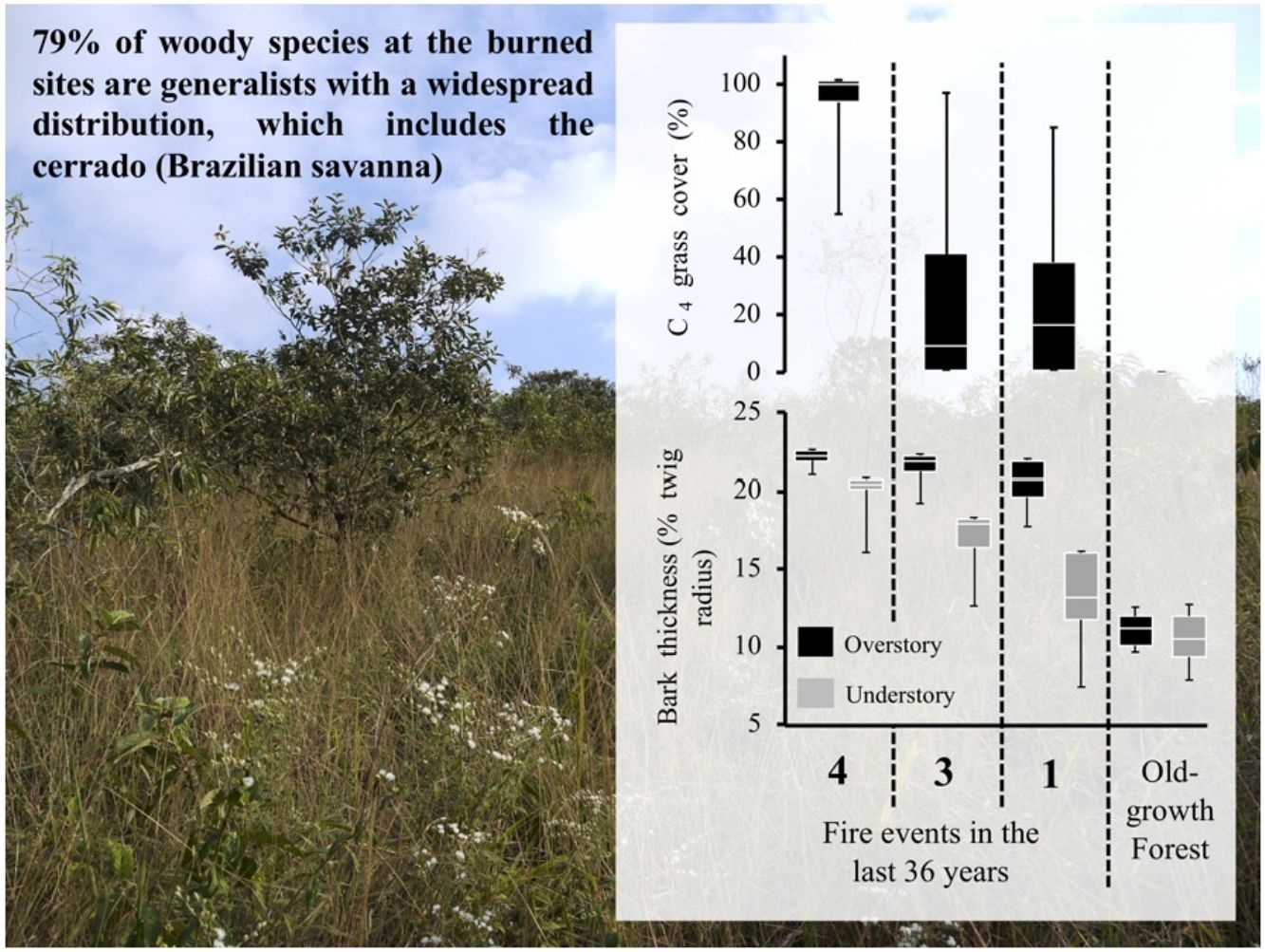
Most tropical forests are threatened by a myriad of human-induced disturbances, associated with land use changes, altered fire regimes and direct deforestation. The combined effect of multiple disturbances can shift forests towards a new, resilient state that is qualitatively distinct in structure, species composition and function. We found that abandoned pastures affected by fires in the Brazilian Atlantic forest showed similarity in terms of vegetation structure and plant functional traits to a savanna-like ecosystem. Burned communities exhibited more C4 grass cover, higher proportion of resprouts and lower canopy cover as compared to the old-growth forest. Moreover, 79% of woody species at the burned sites are generalists with a widespread distribution, which includes the cerrado (Brazilian savanna). Woody species composition was strikingly different from old-growth forests, since burned sites were dominated by Moquiniastrum polymorphum (Less.) G. Sancho (Asteraceae), a fire-resistant species with a ruderal behavior that was absent in the old-growth forests. The results observed in this study provide biological evidence for an arrested succession with the establishment of a savanna-like ecosystem as an alternative stable state. These findings reinforce the notion that the establishment of an alternative stable state (e.g., savanna-like state) hypothesis for the Brazilian Atlantic Forest should be considered, especially under the current climate change scenarios.
Human activities have caused substantial transformations in tropical forests around the world over the last century (Roque et al., 2018). These transformations have led to biodiversity losses, drastic changes in community structure and ecosystem functioning, and, consequently, have affected important ecosystem services provided by tropical forests (Gardner et al., 2007; Shimamoto et al., 2018). In the Neotropics, the interaction between deforestation, disturbance events (e.g., fire) and climate change can drive communities to a large-scale dieback of tropical forests through a savannization process (Nobre et al., 1991; Salazar et al., 2007; Nobre and Borma, 2009; Flores et al., 2016; Staal et al., 2018). Establishment and maintenance of alternative stable states (e.g., savanna-like state) are mainly associated to disturbance regimes (see Murphy and Bowman, 2012; Magnuszewski et al., 2015). Therefore, this process has important consequences for the overall resilience of tropical forests, even more if we consider forest and savanna as alternative stable states that exhibit sharp transitions (Hirota et al., 2011; Staver et al., 2011; Dantas et al., 2016; Van Nes et al., 2018).
The hypothesis of savannization of tropical forests has been predicted on the basis of climate modeling (Salazar et al., 2007; Hirota et al., 2011; Franchito et al., 2012; Anadón et al., 2014), but sound biological evidence is still scarce. According to Staver et al. (2011), climate is not the main factor that explains forest-savanna distribution in the Neotropical region. On the other hand, fire can generate discontinuities on forest distribution and drive communities to alternative states (Staver et al., 2011; Dantas et al., 2016). Observational studies about the effects of land use and repeated fires in eastern Amazonia indicate the occurrence of a process termed “secondarization” (Barlow and Peres, 2008). Secondarization refers to human-induced disturbances in old-growth forests that lead to biodiversity impoverishment and communities dominated by a small set of early successional forest species (Barlow and Peres, 2008; Joly et al., 2014). On the other hand, savannization is mainly associated with aridification, intense land use changes, and fire, leading to drastic changes in vegetation structure and how these communities respond to disturbance (Borhidi, 1988; Silverio et al., 2013). Thus, in order to describe the establishment of a savanna-like state vegetation structure, species composition, and functional traits should to be considered to assess the differences between a savanna and a forest ecosystem (Ratnam et al., 2011). However, the concept of savanna-like adopted in this study represent communities that look like a savanna, but they are not an original savanna (e.g., Ovalle et al., 1996; Gómez Sal et al., 1999)
The same factors (deforestation, conversion into pastures, and recurrent fires) that are related with “secondarization” and “savannization” in the Amazon forest (Nobre et al., 1991; Salazar et al., 2007; Barlow and Peres, 2008) have been occurring in the Brazilian Atlantic forest for at least two hundred years (Joly et al., 2014; Scarano and Ceotto, 2015). There is a massive presence of anthropogenic pastures in this biome (Strassburg et al., 2014) and about 28% of the original forest cover remains, mostly as edge-affected or secondary vegetation fragments (Rezende et al., 2018). Previous studies have demonstrated that heavily disturbed sites can show arrested forest succession (Sansevero et al., 2017), alarming rates of species extinction (Cardoso da Silva and Tabarelli, 2000), and changes in ecosystem function that lead to shifts in species richness and composition (Liebsch et al., 2008; Prieto et al., 2017). Consequently, in order to test the hypothesis that a savanna-like state is taking place in abandoned pastures of the Brazilian Atlantic Forest, we quantified three fundamental attributes that differentiate forest from savanna ecosystems: 1) vegetation structure, 2) woody species composition, and 3) plant functional traits. The study was developed in abandoned anthropogenic pastures subjected to different fire frequencies and in old-growth forests (reference forest ecosystem). This approach can provide fundamental biological evidence of the establishment of alternative stable states and provide empirical support to the notion that the savannization hypothesis should not be overlooked for the Brazilian Atlantic Forest.
Materials and methodsStudy areaThe study was carried out at Poço das Antas Biological Reserve, located in the Rio de Janeiro state, Brazil (22º 32`17``S, 42º 16`50``W). The annual average temperature is 23 ºC with 1900 mm of annual precipitation, with a moderate dry season from June through August (Lima et al., 2006). Vegetation is classified as lowland Atlantic Rainforest (Oliveira-Filho and Fontes, 2000). Small round hills compose the relief, with a mean height of 50 m, and alluvial plains. Ultisols with high Al contents and low P availability are predominant (Lima et al., 2006). Due to previous land use, 48% of the reserve is covered with anthropogenic abandoned pastures and secondary forests (Lima et al., 2006). At least five large-scale anthropogenic fires occurred in the last 36 years. In general, fire starts in private lands and spreads through the reserve, facilitated by an extensive grass cover in abandoned pastures. We selected twelve areas comprising four different land use histories and fire frequencies (High fire frequency – Hi, i.e. burned in 1990, 2002, 2008, and 2010; Intermediate fire frequency – In, burned in 1990, 1993, and 2002; Low fire frequency – Lo, burned only in 1990; and Old-growth forests - Og: no fire events). All sites affected by fire were covered by pastures in 1956 and they were abandoned in 1974 when the Protected Area was implemented. The main grass species observed in these abandoned pastures were Melinis minutiflora P. Beauv., Imperata brasiliensis Trin., and Urochloa mutica (Forssk.) T.Q. Nguyen. (Lima et al., 2006). Land use histories and fire frequencies in each site were determined from aerial photographs, satellite images, technical reports from Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio), and field surveys. Distances of abandoned pastures with different fire frequencies (Hi, In and Lo) from old-growth forests in the landscape range from 70 to 200 m, which means a lack of seed dispersal limitation and high potential forest resilience (e.g., Pereira et al., 2013; Rezende et al., 2015).
Vegetation surveyWithin each land use history class, we delimited three sites, and six plots of 10 m × 10 m were randomly sampled at each site, totalizing eighteen samplings units per area. Both, overstory (plants with diameter at breast height dbh ≥5.0 cm) and understory (1.0 ≤ dbh < 5.0 cm) were surveyed. Understory plants were measured in small sub-plots (5 m × 5 m) located at the left lower corner of each overstory plot facing northwards. Botanical material was collected for species identification. We recorded the geographical distribution of all species present at the burned sites and the 15 most abundant species (50% of total abundance) in the old-growth forest site using the List of Species of the Brazilian Flora (now Flora do Brazil 2020, available in: http://floradobrasil.jbrj.gov.br/2012) (Appendix A in Supplementary material).
Vegetation structureLeaf area index (LAI), C4 grass cover and abundance of resprouting plants were quantified as a measure of community structure response to fire. LAI and grass cover are strongly related to fire dynamics (Bond, 2008; Hoffmann and Franco, 2003), while the capacity to resprout is related to the ability to persist in ecosystems with recurrent disturbances (Bond and Midgley, 2001). LAI was quantified using hemispherical photographs (see Chianucci and Cutini, 2012) with a Nikon Coolpix 4500 camera equipped with Nikon FC-8 fisheye lens. Photographs were taken at the center of each plot one meter above the ground and LAI was calculated using the software Gap Light Analyzer (GLA) (available at http://www.caryinstitute.org/). The percentage of grass cover was visually estimated in two sub-plots of 2 m2 (2 m × 1 m) within each overstory plot (10 m × 10 m). Abundance of resprouting plants is the relative proportion of basal resprouting individuals within each plot.
Functional traitsBark thickness (% of twig bark thickness) and potential height were measured for 142 species, belonging to 86 genera and 38 families (Appendix B in Supplementary material). These traits are informative of plant response to fire (Uhl and Kauffman, 1990; Hoffmann and Franco, 2003) and to differentiate savanna from forest species (Ratnam et al., 2011). Bark thickness was measured with a digital caliper and calculated as a percentage of twig bark thickness radius. Therefore, percentage of twig bark thickness was used as proxy of bark thickness (see Paine et al., 2010). For this functional trait there were in all between one to eight individual plants selected from each species according to species abundance. Potential height was quantified based on field and herbarium specimens from Barbosa Rodrigues Herbarium at Rio de Janeiro Botanical Garden (RB), considering plants of the same species collected only in the Poço das Antas Biological Reserve. This procedure was adopted in order to avoid trait variation caused by large scale changes in environmental conditions (e.g., temperature, rainfall, soil, etc).
Data analysisTo verify whether areas differed in vegetation structure (LAI, C4 grass cover and abundance of resprouting plants) and functional traits (bark thickness and potential height), we performed a permutation-based analysis of variance (Pillar and Orlóci, 1996), using 10,000 permutations. This analysis was chosen because it works well with univariate response data (see Carlucci et al., 2015) and because it makes no assumptions about normality (Pillar and Orlóci, 1996). A previous study (Sánchez-Tapia, 2011) with the same sampling units used here found a minor role for spatial autocorrelation in explaining community data, and we thus did not control for spatial autocorrelation in the subsequent analyses. We considered each plot as a replicate and groups of six plots (within each area) as a block. Thus, we tested the differences between areas (by contrasts) controlling for internal variation (within blocks). For each functional trait, the community-weighted mean was calculated for each plot. Statistical analyses were made separately for each vegetation layer (overstory and understory). Differences in species composition in the overstory was described with non-metric multidimensional scaling (NMDS) (Legendre and Legendre, 2012). All statistical analyses were performed in the MULTIV program (Pillar, V. v 2.3. available in: http://ecoqua.ecologia.ufrgs.br)
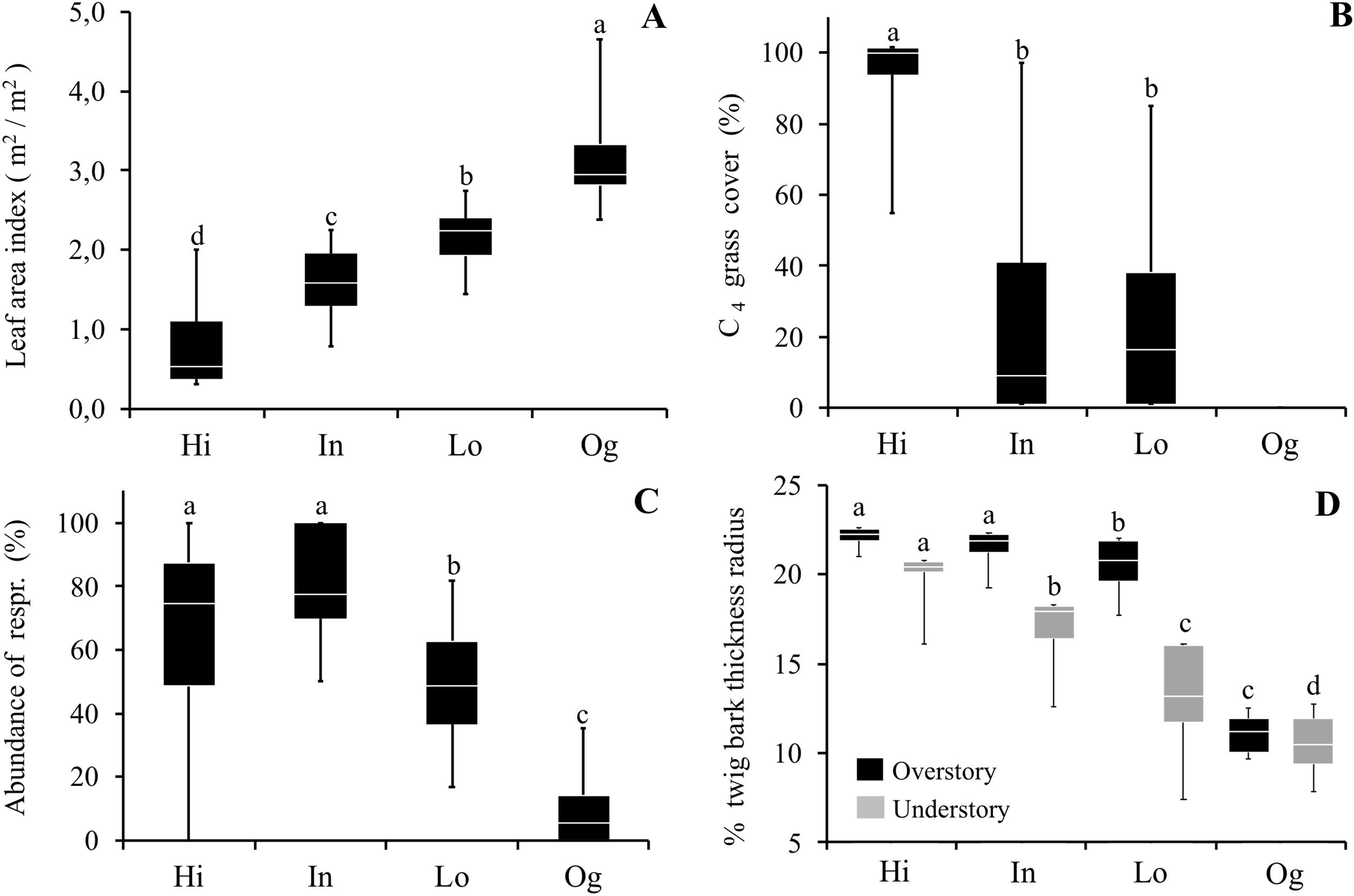
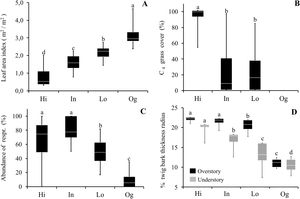
ResultsOur results reveal that plant communities subjected to fire exhibited changes in vegetation structure and functional traits more compatible with cerrado (Brazilian savanna) rather than to old-growth forests. Considering the vegetation structure, the abundance of resprouting individuals and the C4 grass cover was higher in burned sites, while leaf area index was lower, as compared to the old-growth forest (Fig. 1). Grass cover was negatively related to leaf area index (LAI, R2 = 0.77 p < 0.0002). Even in sites with intermediate and low fire frequencies (20 years since the fire event), mean values of C4 grass cover were 22% and 23%, respectively, but some plots presented 90% of grass cover (see maximum values in Fig. 1). High C4 grass cover in burned sites indicates that there is a susceptibility to further fire events. Communities subjected to fire also showed a high abundance of resprouting plants (Hi: 67%; In: 79%; Lo: 49%) (Fig. 1C). Regarding the functional component, communities subjected to fire had higher bark thickness (Fig. 1) and lower potential height. Except for Hi and In, all sites presented significant differences from each other in potential height (Hi: 8 m; In: 7.8 m; Lo: 8.7 m Og: 14 m p < 0.001). Therefore, communities subjected to fire had greater bark thickness and C4 grass cover, and lower-statured trees with a more open canopy (see Fig. 2A).
Comparisons of vegetation structure and functional traits between areas affected by fire and old-growth forests. (A) Leaf area index; (B) C4 grass cover; (C) Abundance of resprouting individuals; and (D) % twig bark thickness in the overstory (plants with the diameter at breast height ≥5.0 cm – black boxes) and understory (diameter at breast height 1.0 ≤ dbh <5.0 cm – gray boxes) in areas with different fire frequencies (Hi: 4 times; In: 3 times; Lo: 1 time) and an old-growth forest (Og). Boxes represent 25–75 percentiles, lines within boxes represent the median value, and bars indicate the minimum and maximum values. Different letters indicate significant differences at p < 0.01.
Vegetation structure and functional traits of the dominant tree species in areas affected by fire in the Brazilian Atlantic forest. (A) Site with high fire frequency showing high C4 grass cover and low leaf area index; (B) A stem of M. polymorphum – Asteraceae with its thick bark. This photograph was taken one year after the last fire event. (C) Resprouting of the most abundant tree species in burned areas – Moquiniastrum polymorphum (Less.) G. Sancho – Asteraceae.
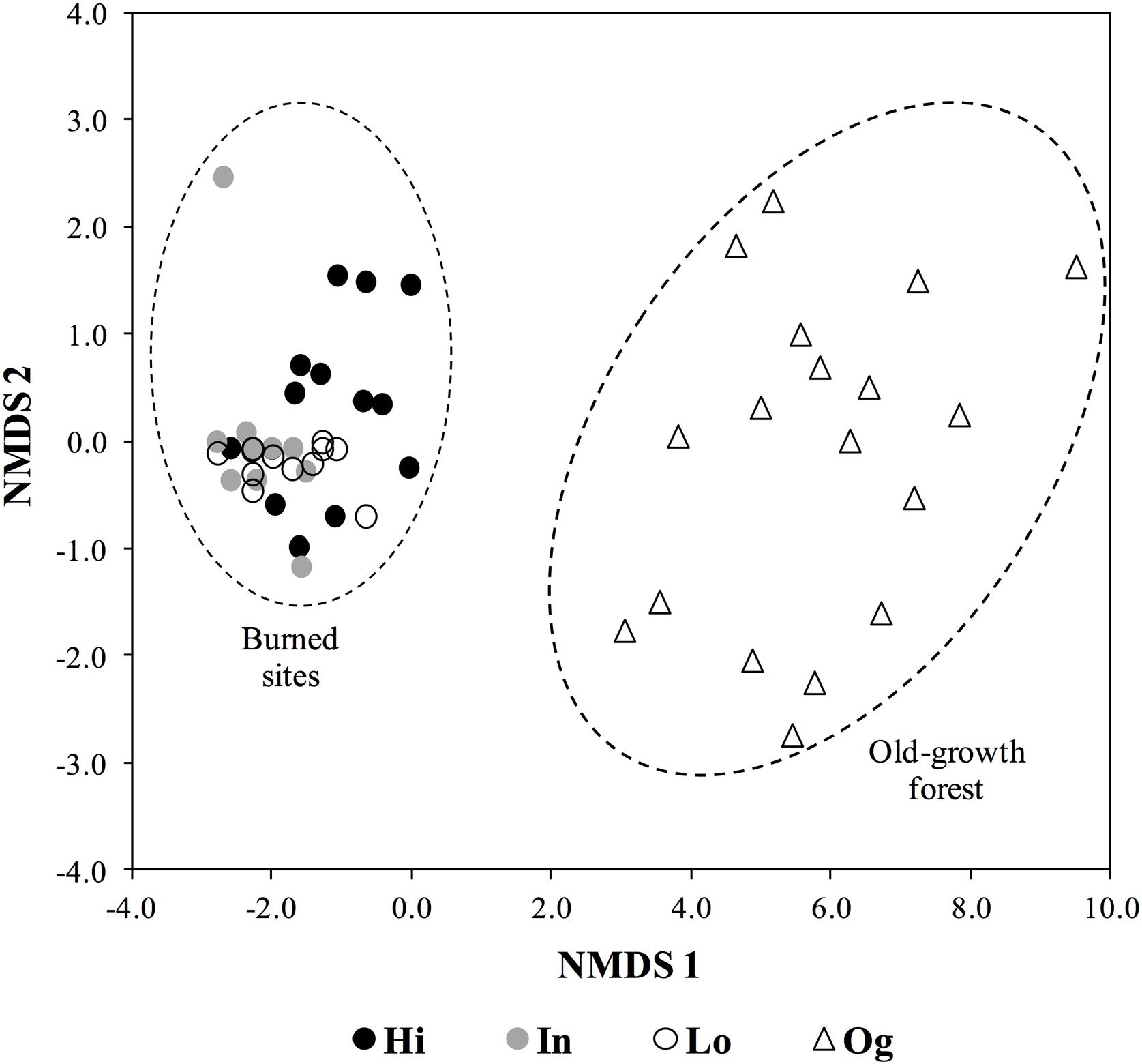
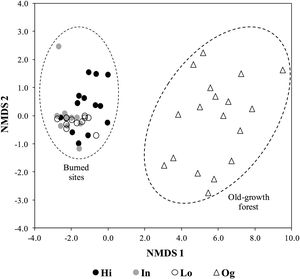
Lastly, when considering floristic composition, 79% of woody species at the burned sites are generalist that show a widespread distribution, which include the cerrado (Brazilian savanna) (Appendix A in Supplementary material). Moreover, of the 15 most abundant species in the old-growth forest, representing 50% of total abundance, ten species are endemic to the Atlantic forest, while only three also occur in the cerrado (Appendix A in Supplementary material). Species composition at the community level was strikingly different to old-growth forests, irrespective to fire frequency (Fig. 3). Burned sites were dominated by Moquiniastrum polymorphum (Less.) G. Sancho (Asteraceae) (Fig. 2), which was absent in the old-growth forests. Therefore, considering the main differences between forests and savannas, the two components analyzed (vegetation structural and functional traits) confirm the establishment of a savanna-like state in abandoned pastures subject to fire in the studied sites of Brazilian Atlantic Forest.
Non-metric multidimensional scaling (NMDS) ordination of abundance of overstory plants in communities subject to different fire frequency (High fire frequency – Hi, i.e. burned in 1990, 2002, 2008, and 2010; Intermediate fire frequency – In, burned in 1990, 1993, and 2002; Low fire frequency – Lo, burned only in 1990) and old-growth forest (Og) in the Brazilian Atlantic forest.
This study provides biological evidence for the establishment of a savanna-like state in abandoned pastures in the Brazilian Atlantic Forest biome. These results also pave the way for further studies to test the savannization hypothesis in a broader spatial and temporal scales, similar to those developed in other neotropical forests (Borhidi, 1988; Cavelier et al., 1998; Silverio et al., 2013). However, and in contrast to other studies in which not all components analyzed suggest the savannization hypothesis (e.g., Veldman and Putz, 2011; Silverio et al., 2013), we found that vegetation structure and functional traits support it. The analysis of these components altogether is also a key factor that differentiates our findings from the process termed “secondarization”, observed in eastern Amazonia (Barlow and Peres, 2008). We suggest that the main factor associated with this process is a long history of land use and degradation that led to increased fire frequencies. Recurrent fires can lead to a bottle-neck effect increasing juvenile mortality (see Hoffmann et al., 2012), reducing tree cover, increasing grass cover, and consequently favoring the occurrence of new fire events. These results suggest that the occurrence of fire is a key factor in the establishment of a savanna-like state observed in these sites.
Fire regime and C4 grass cover are directly related to each other (Bond, 2008; Hoffmann et al., 2012). C4 grasses have higher productivity and lower decomposition rates than C3 plants, which contributes to accumulation of flammable fuel loads (Bond, 2008). The relationship between grass cover and low leaf area index is an important threshold, known as a fire-suppression threshold, that regulates fire events in savanna communities (Hoffmann et al., 2012). Our findings demonstrate that the changes observed in the burned sites agree with the category of savanna according to the classification proposed by Ratnam et al. (2011): they have an open canopy and present a high abundance of C4 grass species (Melinis minutiflora P. Beauv. and Imperata brasiliensis Trin.). It is important to stress that M. minutiflora is an exotic invasive species, introduced by cattle breeders, which has become equally noxious and competitive in old-growth cerrado communities (Hoffmann et al., 2004). With regards to the tree layer, the dominant tree species in the abandoned pastures (Moquiniastrum polymorphum (Less.) G. Sancho – Asteraceae) is a common species in dry forests and cerrado (Durigan, 2006; Oliveira-Filho, 2006; Cava, 2019), as well as in abandoned anthropogenic pastures in the cerrado (Cava, 2019). The abundance of M. polymorphum in sites subjected to fire may be explained by its fire resistance (Brandes et al., 2018) and seed dispersal capacity in human-modified landscape (Jesus et al., 2012).
Bark thickness was positively correlated with fire frequency. This is one of the most important plant traits in avoiding fire-induced tree mortality (Hoffmann et al., 2012). Forest species are known to have lower values of bark thickness when compared to savanna species (Uhl and Kauffman, 1990). From an evolutionary perspective, the appearance of species with high bark thickness in the savanna occurred as an adaptive response to fire and it also coincided with the expansion of C4 grasses in this biome (Simon and Pennington, 2012). In the overstory, the community-weighted mean of bark thickness of burned communities was observed to be twice as large as in the old-growth forests. In the same way, resprouting plants were abundant at burned sites, which can be interpreted as a strategy for persistence in response to a disturbance such as fire (Bond and Midgley, 2001). Moreover, such abundance was much higher than that of secondary forest in the Atlantic Forest, where less than 10% of the individual originate from resprouting (Simões and Marques, 2007). Therefore, functional similarities between the species present in the burned sites and those typical from the savanna reinforce the need to consider the savannization hypothesis in abandoned pastures affected by fire.
Despite the fact that our results are restricted to one region, we consider that it can be generalized to other areas with similar land use history in the Brazilian Atlantic Forest, given the degradation history of this biome. A study about global forest resilience based on tree cover and precipitation showed that several areas of Brazilian Atlantic Forest, which include our study area, have low forest resilience and a high probability to turn into a savanna or a treeless state (Hirota et al., 2011). A recent study also showed that soil erosion plays a role in reducing ecological resilience, which combined with fire events can also accelerate ecosystem shifts (Flores et al., 2019). This change observed in the present study does not result into an old-growth savanna but, on the contrary, the outcome is a derived savanna-like ecosystem (sensuVeldman, 2016). Consequently, the extent and magnitude of the likelihood of savannization of the Atlantic Forest requires urgent attention.
Our results have important practical implications because differences between forests and savanna-like ecosystems involve important changes in the ecosystem services provision, their responses to disturbance and climate change. In our case, biodiversity conservation is also compromised, since the derived-savanna showed low woody species richness and with the presence of invasive alien species in the grass layer. Our findings reveal a worrying future for the Atlantic Forest because the ongoing climate change is likely to exacerbate the observed savannization (e.g., Franchito et al., 2012; Anadón et al., 2014) and indicate that its transformation into savanna-like might be a matter of time, a time that would be particularly short under the most likely climate change scenarios.
Conflicts of interestNone.
We thank Carlos Nobre and Toby Gardner for valuable comments on an earlier version of the manuscript. This research was partly funded by PROBIO II (BIRD/MMA/MCTI/JBRJ). We are very grateful to Adilson Pintor and Antonio Tavares for fieldwork support. Sansevero, J.B.B. received a PhD scholarship from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and the Programa de Doutorado Sanduíche no Exterior (PDSE - CAPES).