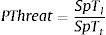The assessment of species conservation status traditionally relies on population data. In the absence of such data, biological attributes have been applied to predict the degree of species’ vulnerability. Our study investigated the accuracy of biological attributes to predict species’ vulnerability to extinction by contrasting matches and mismatches between threat predictions and the conservation status of reef fish species in the Brazilian Red List of Threatened Species. We further analyzed the patterns of distribution of threatened species under different attribute categories. The proportion of matches were higher for Elasmobranchii (92%), habitat specialists (88%) and highly mobile species (81%). Also, other attributes as macrocarnivorous and mobile invertivore-based diets, habitat intermediate-generalists, and large body size had over 70% of match in predictions. Fifty-six Brazilian reef fish species share these attributes but are not listed as threatened, which makes them even more vulnerable to future extinctions. Higher proportions of threatened species presenting these biological attributes were found in southeast Brazil (states of Espírito Santo and São Paulo). Our study has revealed that biological attributes may serve as an alternative tool to predict reef fish species vulnerability.
We are living the Anthropocene, a geological period marked by profound human-derived impacts to planet Earth, its biodiversity and natural ecosystems (Young et al., 2016). As a consequence, growing extinction rates have reached multiple taxonomic groups both at sea and on land (Dirzo et al., 2014; McCauley et al., 2015). Despite being in a more incipient stage, defaunation in the marine realm already presents profuse effects and is likely to accelerate in the next years (Young et al., 2016). Coral reef ecosystems harbor fascinating species richness, including one the most diverse vertebrate assemblages on Earth: reef fishes. Reefs are worldwide threatened by a combination of human-derived impacts such as pollution, overexploitation, habitat loss and climate change (McCauley et al., 2015; Hughes et al., 2017). In particular, fishing is the most pervasive and ancient cause of disturbance to marine ecosystems (Jackson et al., 2001). Despite the increasing impacts on reef ecosystems, only 47 reef fish species are listed as globally threatened with extinction (Parravicini et al., 2014). If small-ranged species were considered as threatened, since the International Union for the Conservation of Nature (IUCN) criteria predicts this extinction risk (criteria VU D2 ‘Very restricted distribution and plausibility and immediacy of threat’; IUCN, 2019) this number would rise to 779 fish species (Parravicini et al., 2014). In the Southwestern Atlantic, recent data show that 78 reef fishes are threatened with extinction, representing 10.6% of the regional reef fish fauna (Pinheiro et al., 2018). Along the Brazilian biogeographical province, 98 reef fish species are listed as threatened (MMA, 2018) highlighting the risk of biodiversity loss at this scale. Brazilian reefs are threatened by the impacts deriving from the growing human population, this being associated mainly with overfishing and pollution, causing changes in reef structure and functioning (Leão et al., 2019). Changes in reef complexity (e.g. coral bleaching) and the removal of fish species in particular trophic levels, such as top predators (fisheries target species) and herbivores are some of the effects associated to anthropogenic impacts on reefs (Pinheiro et al., 2010; Bender et al., 2014; Leão et al., 2019).
Red lists of threatened species, a fundamental conservation tool, indicate population trends for different taxa and reveal the major impacts imposed to global biodiversity. The IUCN provides an evaluation system based on quantitative criteria to assess the conservation status of species (IUCN, 2019). The set of criteria uses current population size and/or trends and distributional range (extent of occurrence and area of occupancy) under properly identified impacts to indicate species extinction risk. However, appropriate population data are not available for the majority of species, hampering the assessment of many organisms under such IUCN criteria (Rodrigues et al., 2006). Specifically, the marine realm is poorly covered in the IUCN Red List, representing only 15% of species assessed (IUCN, 2019). The first IUCN Red List assessments were focused on selected species (as mammals and birds) that could attract conservation efforts. IUCN Specialist Groups have contributed to comprehensively assess several taxonomic groups across terrestrial and marine environments. Yet red lists are still biased by scientific knowledge (Rodrigues et al., 2006), and more investment is needed in evaluations of marine species applying IUCN criteria, both at global and regional levels. In this context, alternative methods may provide insights to predict extinction vulnerability (Bender et al., 2013).
Surveys on species vulnerability to extinction have focused on attributes such as patterns of species distribution (Hawkins et al., 2000), their degree of ecological specialization (Graham et al., 2011), life-history attributes (Cheung et al., 2005) and biological attributes (Bender et al., 2013). Conversely, data on declining trends can help identify attributes associated with species vulnerability. Body size, occupancy and rarity may indicate declines that will potentially lead to global extinction, such as in small-bodied animals and food or habitat specialists that tend to be more vulnerable (Graham et al., 2011). For fish species, attributes such as late maturity and slow growth, which are correlated with body size (Reynolds et al., 2005), are proxies of the vulnerability of species that are not yet threatened with extinction (Kotiaho et al., 2005). Attributes can also signal vulnerability to climate change, which in reef fishes is related to diet and habitat specialization, larval settlement and body size (Graham et al., 2011). Body size is considered a useful tool for detecting skates and ray species at risk, possibly due to its correlation with mortality rates and age at maturity (Dulvy and Reynolds, 2002).
Combining biological attributes (maximum body size, trophic category, reproductive attributes) and major threats (fishing impact, major fisheries target), Bender et al. (2013) predicted the extinction risk for 599 reef fish species from the Brazilian Province. In their paper, the threat probability represents the chance of reef fish species being threatened with extinction considering their set of biological attributes and current threats. This study anticipated the publication of the Brazilian Red List of Threatened Species (Brasil, 2014). The Brazilian list has applied IUCN regional criteria to evaluate over 1376 freshwater and marine taxa and lists 98 reef fish species as threatened with extinction. Here, we tested the accuracy of biological attributes in predicting the extinction risk of reef fishes from the Brazilian Province. We compared the probability of extinction calculated by Bender et al. (2013) – from biological attributes and major threats – with the threat categories for fish species from the Brazilian Red List of Threatened Species. We investigate the number of matches and mismatches between different methods and reveal which attributes can be applied to predict species vulnerability to extinction. We further analyzed the distribution of threatened species and its biological attributes along the Brazilian Province and offer recommendations into reef fishes management and conservation.
Material and methodsDatabaseTwo databases were used in our study. The first one comprises occurrences of reef fish species (Bender et al., 2013) along the Brazilian Province. Also, the database contains biological attributes (e.g. maximum body size, trophic category, maximum depth), the conservation status at global and national level, and the probability of extinction for each species. In Bender et al. (2013), the extinction probability was expressed as a proportion, ranging from 0 to 100%. This value was estimated for each species and represents the probability of a species being threatened according to a combination of its biological attributes and the major threats. Species listed with a threat probability greater than 40% were considered highly vulnerable (sensuBender et al., 2013). The second database corresponds to the updated list of Southwestern Atlantic reef fishes, including 733 species (Pinheiro et al., 2018). We updated the categories of species’ conservation status in this database, according to the current Brazilian Red List of Threatened Species (Brasil, 2014, 2018) . Threatened species were considered those categorized as Critically Endangered (CR), Endangered (EN) and Vulnerable (VU). Species categorized as Data Deficient (DD) and Non-evaluated species (NE) were not considered in our study.
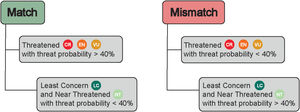
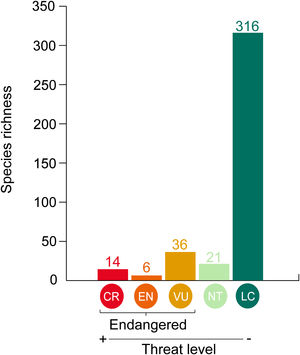
Data analysisWe considered species listed both in Bender et al. (2013) and Pinheiro et al. (2018), totalizing 512 reef fish species: 464 Teleostei and 48 Elasmobranchii. We contrasted the threat probability for reef fishes in Bender et al. (2013) to their status according to the Brazilian Red List of Threatened Species. The proportions of match and mismatch predictions were calculated (Fig. 1). Match predictions were those where (i) species listed as threatened (CR, EN and VU) in the Brazilian Red List with a threat probability greater than 40% in Bender et al. (2013); (ii) species listed as Near Threatened (NT) and Least Concern (LC) with a threat probability smaller than 40% in Bender et al. (2013). The predictions considered mismatches were the following: (i) species listed as threatened (CR, EN and, VU and NT) in the Brazilian Red List with a threat probability smaller than 40% in Bender et al. (2013); and (ii) species categorized as LC and NT but with a threat probability greater than 40%.
Matches and mismatches between the predictions made by Bender et al. (2013) and the Brazilian Red List of Threatened Species. Conservation status: CR – Critically Endangered; EN – Endangered; VU – Vulnerable; NT – Near Threatened; and LC – Least Concern.
From the proportions of matches and mismatches in predictions, we explored which biological attributes were more frequently associated with the match predictions.
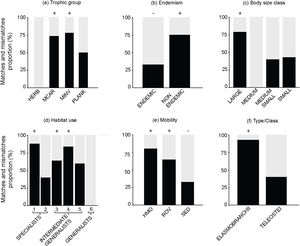
The following biological attributes were analyzed for the proportions of matches and mismatches in each attribute category: trophic group (macrocarnivores, mobile invertivores, sessile invertivores, herbivores, omnivores and planktivores); body size class (small: <10 cm; medium-small: 10.1−25 cm; medium: 25.1−50 cm; large: >50 cm); endemism (endemic or non-endemic to the Brazilian Province); type/class (Teleostei and Elasmobranchii); mobility (high mobility, roving mobility, sedentary) and habitat use (specialists, intermediate generalists and generalists). Habitat use categories were based on the number of habitats that each species is associated to, according to the literature: specialists use 1–2 habitats; intermediate-generalists are found in 3–5 habitats and generalists are associated to 6–7 different habitats (Pinheiro et al., 2018). To test the statistical significance between match and mismatch proportions in each attribute category we used a two-tailed Binomial test (match proportion ≠ mismatch proportion; p-value < 0.05).
We then explored the distribution patterns of threatened species according to their biological attributes. A set of distribution proportion maps was created in 21 sites along the Brazilian Province: states of Santa Catarina (SC), Paraná (PR), São Paulo (SP), Rio de Janeiro (RJ), Espírito Santo (ES), Alagoas (AL), Bahia (BA), Paraíba (PB), Pernambuco (PE), Rio Grande do Norte (RN), Ceará (CE), Pará and Maranhão (PA-MA); Ilha Grande Bay (Igr), Arraial do Cabo region (ArC), Abrolhos (Abr), Trindade- Martin Vaz insular complex (Tri), Vitória-Trindade chain (VTC), Manuel Luís Parcel (PML), São Pedro e São Paulo Archipelago (SpSp), Fernando de Noronha Archipelago (FN) and Rocas Atoll (ROC). In each site, the proportion of threatened species was calculated as the ratio between species richness and the total number of threatened species in each attribute.
The proportion of threatened species (PThreat) in each biological attribute was calculated with the following formula:
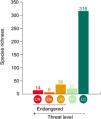
Where SpT is the number of threatened species for a given attribute category in site (l) and the total number of species along the Brazilian Province (t). For instance, a total of 39 threatened species are categorized as large-bodied in the Brazilian Province. In ROC, 11 threatened species are large-bodied. To calculate the proportion of threatened species with large body sizes at ROC, we divided the number of threatened species within this category (n = 11) by the total number of threatened species for this attribute (n = 39). Using this rationale, we mapped the distribution of threatened species in the following attribute categories: large body size; mobile invertivores and macrocarnivores; Teleostei and Elasmobranchii, and highly mobile species. Species occurrence records were extracted from the Southwestern Atlantic Reef Fish database (Pinheiro et al., 2018). We also mapped the richness of threatened species within different attribute categories in each site (Supplementary Fig. S1). To test whether sites along the Brazilian Province present similar structure regarding threatened reef fish species, we applied a Cluster analysis in the proportions of threatened species for the following attributes: diet, habitat use and body-size classes. Clusters were built through the “average” agglomerative method and p-values at cluster nodes were measured by bootstrap resampling (1000 replicates) (see Supplementary Fig. S2). All analyses were performed using the R software version 3.4.4 (R Core Team, 2018).ResultsOut of 512 evaluated reef fish species, 56 were listed as threatened with extinction according to the Brazilian Red List. Of these species, 26% (n = 14) were listed as CR, 10% (n = 6) as EN, 64% (n = 36) as VU (Fig. 2). The comparison between the proportions of match and mismatch predictions for trophic group categories has revealed that match proportions were significantly greater for mobile invertivores (78%; p < 0.001) and macrocarnivores (74%; p < 0.001) relative to other categories (Fig. 3a). For other evaluated attributes, significant categories for match predictions – those where species listed as threatened also present threat probability >40% in Bender et al. (2013) – were non-endemics (76%; p < 0.001) (Fig. 3b) and large body size (79%; p < 0.001) (Fig. 3c), whereas endemic species showed the greater mismatch proportions (33%; p < 0.001). For habitat use, significant categories included specialists (88%; p < 0.001) and intermediate generalists (63% e 83%, respectively; p < 0.001) (Fig. 3d). For fish mobility, the highest proportions of match predictions were identified for highly mobile (81%; p < 0.001) and vagrant species (65%; p < 0.05), whereas sedentary species had the greater mismatch proportion (33%; p < 0.001) (Fig. 3e). For type/class, Elasmobranchii (92%; p < 0.001) showed the highest match proportion (Fig. 3f).
Proportion of match (black) and mismatch predictions (gray) for each biological attribute. (a) Trophic group: MCAR – macrocarnivores; MINV – mobile invertivores; HERB – herbivores; PLANK – planktivores; (b) Endemism; (c) Body Size class; (d) Habitat use: 1,2 – specialists; 3,4,5 – intermediate-generalists; 6 – generalists; (e) Mobility: HMO – high mobility; ROV – roving mobility; SED – sedentary; and (f) Type/Class. +/− Values denote significant difference at p < 0.05 with the Two-Tailed Binomial test. +indicate good predictors while – indicate poor predictors.
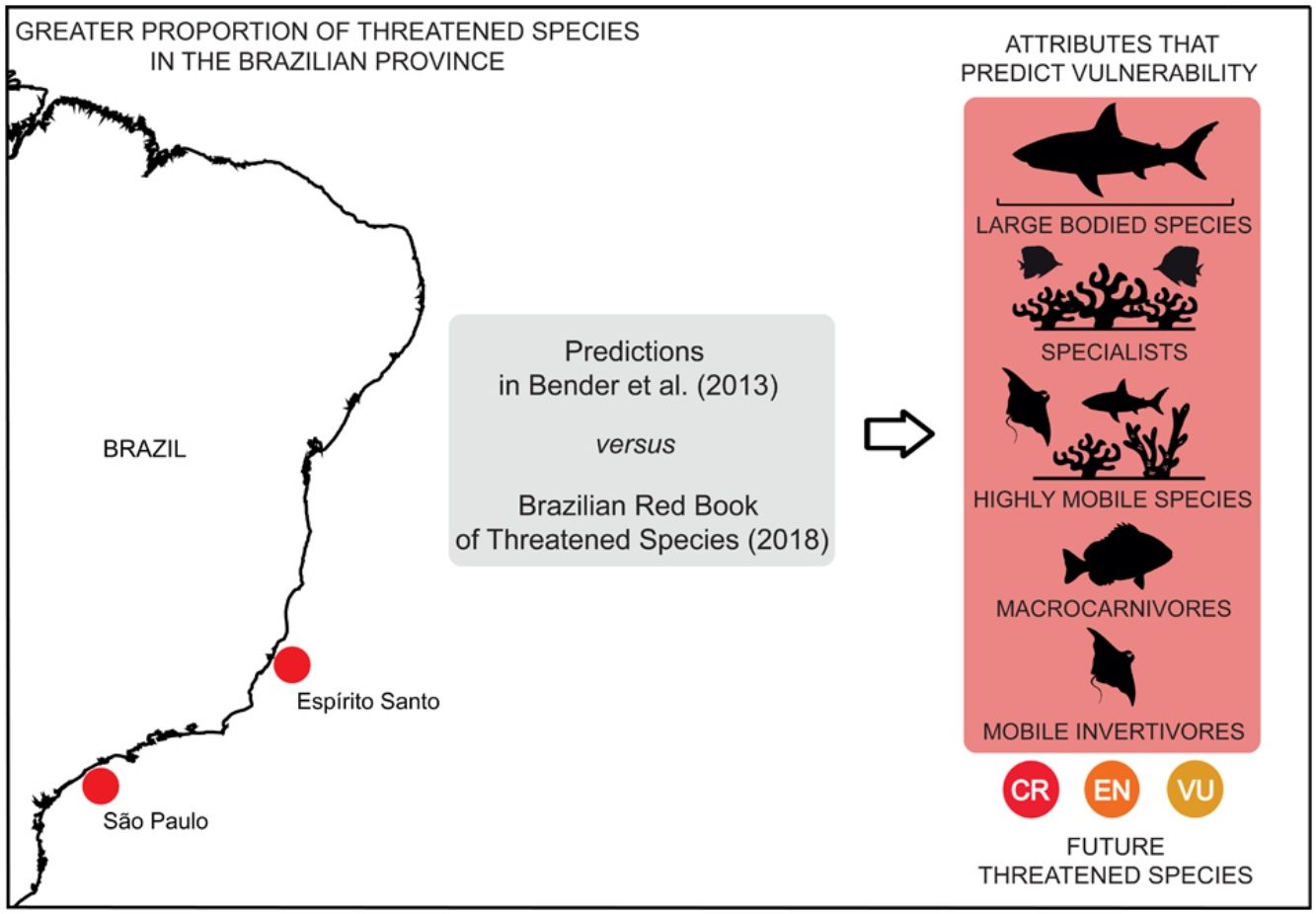
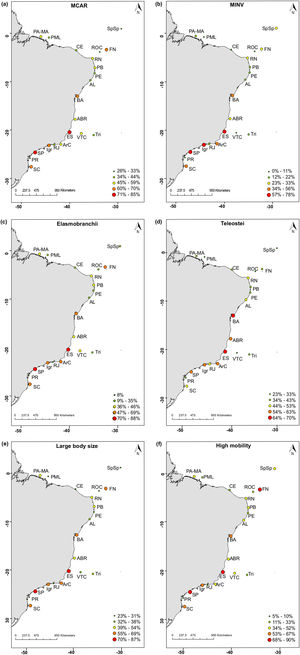
The distribution of threatened species has revealed that there is a greater proportion of threatened species in Espírito Santo (ES) and São Paulo (SP). This pattern was observed for the following attributes: macrocarnivores and mobile invertivores (Fig. 4a, b), Elasmobranchii (Fig. 4c), large-bodied (Fig. 4e) and highly mobile species (Fig. 4f). High proportions of threatened Teleostei (Fig. 4d) have been observed in Espírito Santo (ES) and Bahia (BA). Ilha Grande (Igr), in the coast of Rio de Janeiro State, also concentrates a high proportion of threatened invertivores (Fig. 4b) and Fernando de Noronha (FN), of highly mobile species (Fig. 4f). Maps for the distribution of threatened species richness along Brazilian reefs revealed the same patterns as those identified for mapped proportions (Supplementary Fig. S1). Overall, the greater proportions of threatened fish species are found in southeastern reefs along the Brazilian coast.
The distribution of threatened reef fish species proportion in different biological attribute along the Brazilian Province. (a) Macrocarnivores (MCAR); (b) Mobile invertivores (MINV); (c) Elasmobranchii; (d) Teleostei; (e) Large-bodied species; (f) Highly mobile species. The color gradient represents the percentage of threatened species with each attribute. Sites: SC, state of Santa Catarina; PR, state of Paraná; SP, state of São Paulo; RJ, state of Rio de Janeiro; Igr, Ilha Grande Bay; ArC, Arraial do Cabo; ES, state of Espírito Santo; Abr, Abrolhos; BA, state of Bahia; Tri, Trindade-Martin Vaz insular complex; VTC, Vitória-Trindade chain; AL, state of Alagoas; PB, state of Paraíba; PE, state of Pernambuco; RN, state of Rio Grande do Norte; CE, state of Ceará; PA-MA, states of Pará and Maranhão; PML, Manuel Luís Parcel; SpSp, São Pedro and São Paulo Archipelago; FN, Fernando de Noronha Archipelago; ROC, Rocas Atoll.
Firstly, it is important to highlight that the work of Bender et al. (2013), indicating Brazilian reef fish with greater vulnerability to extinction, despite having been published before the Brazilian Red List of Threatened Species (Brasil, 2014), had absolutely no influence on species assessments. The Brazilian Red List has been built upon evaluations of species following IUCN regional criteria (Gärdenfors et al., 2001), using species’ current population size and/or population trends and distributional range. Furthermore, specialists from research institutes across the country contributed to species assessments in a participatory and scientifically rigorous process. Thus, species extinction risk based on attributes and major threats (Bender et al., 2013) were not applied to assign fish species to threat categories.
Our study indicates which biological attributes can accurately predict the extinction risk for Brazilian reef fish species. Greater extinctions risks were verified in species with large body sizes, macrocarnivores and mobile invertivore diets, specialists and intermediate generalists, vagrant and highly mobile species and/or belonging to the Elasmobranchii class. These attributes characterize megafauna species (e.g. sharks, rays, mammals and turtles), many of them threatened by anthropogenic activities (McCauley et al., 2015). Megafauna species share life-history features (e.g. low fecundity, late age at maturity, and slow growth) that result in low biological productivity and increase their vulnerability to extinction (Dulvy et al., 2014). In addition, large predators such as groupers, sharks and rays, are commercially valuable and many species have depleted stocks due to overfishing (Sadovy de Mitcheson et al., 2013; Dulvy et al., 2014). Overfishing compromises stock recovery because species with slow growth and late maturity take many generations to regain their population size. Also, the unsustainable removal of these species can trigger cascading effects on lower levels of the food web and change considerably the size spectra (abundance-body mass relationships) compromising the structure and functioning of marine communities (Baum and Worm, 2009). Such effects may impair the resilience capacity of entire marine ecosystems.
Trophic category and body size were investigated in previous studies and showed similar results. Large body sizes (Dulvy and Reynolds, 2002) and a macrocarnivore diet (Bender et al., 2013) were also good predictors of the extinction probability of fish species. Our results revealed that mobile invertivores were also related to match predictions. This is possible because most of these threatened species are rays that feed on mobile invertebrates, such as guitarfishes, sharing life-history features that render populations less resilient to overexploitation (Moore, 2017). Large-bodied rays, such as skates and giant rays, tend to decline over time due to overfishing and correlations with their life-history features (Dulvy et al., 2014).
Habitat use can also be employed as a predictor of species’ vulnerability (Hawkins et al., 2000). Specialist species face greater extinction risk because they are more susceptible to local disturbances and habitat loss (Hawkins et al., 2000; Graham et al., 2011). Also, specialist fish are more dependent on habitat availability when compared to generalists that use a variety of habitat uses. Thus, changes in habitat availability may have a greater impact on the abundance of specialized species (Munday, 2004). Among coral reef fishes, species associated with live coral are at greater risk due to disturbance than are generalists, being more vulnerable to extinction (Wilson, 2008). Therefore, increasing levels of habitat loss and fragmentation generate greater conservation concerns for these species.
Mismatch proportions revealed that endemism and/or sedentary attributes are poor predictors of extinction risk for Brazilian reef fishes. For endemic species, this is possibly an outcome of the large proportion of non-endemic species that are currently threatened with extinction. Endemic species might be imperiled by their small geographic ranges, some also being habitat specialists and locally rare (Hawkins et al., 2000). These species are the most affected by reef degradation, increasing their extinction risk to local disturbances (Hawkins et al., 2000). One hundred and two (102) reef fish species are endemic to the Brazilian Province, 36 being restricted to oceanic islands (Pinheiro et al., 2018).
The distribution of threatened species’ attributes varies along the Brazilian coast and oceanic islands. The northern coast of Bahia holds a large proportion of threatened Teleostei. This region comprises the largest hotspots of reef fish species richness, target species richness and endemism of the Brazilian coast (Vila-Nova et al., 2014), yet this diversity is not effectively protected. No-take marine protected areas (MPAs) are absent and biogenic reefs are poorly protected. Teleostei macrocarnivore species are commercially important in most of the studied sites. For instance, in Abrolhos Bank, groupers and snappers represent 35% of the catches (MPA, 2013). However, at this site, between 55% and 62% of macrocarnivore species are threatened and there are increasing evidence of stocks’ overexploitation (Zapelini et al., 2019). A similar overexploitation pattern has been identified throughout the entire Brazilian Province (Reis et al., 2016). Management initiatives have not been enough to change this worrying scenario. The Brazilian system of MPAs fails to conserve commercially important reef fishes mainly because the poor representativeness of well-designed no-take areas (Magris et al., 2013), lack of resources to manage the existing MPAs (Gerhardinger et al., 2011) and lack of fisheries management and compliance (Gasalla, 2011).
Sharks and rays, several being large-bodied and macrocarnivore species, presented similar spatial patterns in our maps of threatened species distribution. Higher proportions of these attributes were found in the states of ES and SP, where the shark fisheries effort is proportionally higher (Barreto et al., 2017). Overall, the patterns in threatened species attribute distribution are largely driven by the reef fish species richness gradient along the Brazilian Province (Pinheiro et al., 2018), with Espírito Santo concentrating a high proportion of threatened taxa. The coast of Espírito Santo has been identified as one of the most critical areas for reef fish conservation in Brazil (Vila-Nova et al., 2014). Despite being a conservation hotspot, there is only one no-take MPA in the coast of Espírito Santo and the region has suffered with multiple impacts such as the massive dam breach of Mariana (Fernandes et al., 2016). Moreover, the large spacing between Espírito Santo MPAs and those in neighboring states compromises connectivity, and therefore, MPA effectiveness (Vila-Nova et al., 2014).
Non-threatened sharks, jacks and Thunnus spp., for instance, have attributes that can accurately predict their vulnerability to extinction in the near future (see Supplementary Table S1). These attribute combinations include high mobility, a macrocarnivore diet and large body sizes. Shark species such as Carcharhinus acronotus, C. leucas, C. limbatus, and Galeocerdo cuvier, categorized as Near Threatened on the Brazilian Red List, are frequently caught as bycatch and sold in fishery markets in northeastern Brazil (Martins et al., 2018). Additional threats to shark species include overexploitation and catches in nursery habitats and MPAs (Rosa and Gadig, 2014). Generally, these areas are in coastal zones, which overlap with intense fishing activities. The absence of proper species identification both in artisanal and industrial fisheries and the mislabeling of shark meat in markets are other problems for shark conservation (Bornatowski et al., 2014). Fisheries in Brazil have been mostly targeting high-trophic level species and show a recent increasing trend due to the development of offshore fisheries for species such as tuna fish (Vasconcellos and Gasalla, 2001). Thunnus albacares, for instance, represented 28% of longline fisheries in Brazil (1958–2017) (ICCAT, 2019). Seven non-threatened species listed in the Brazilian Red List exhibit declining population sizes at the global scale (Supplementary Table S1). It is important to observe the population trends and monitor non-threatened species that have attribute combinations that are good predictors of extinction vulnerability.
Attempts by part of the Brazilian government (Ministry of Agriculture) and the industrial fishing sector to invalidate the Brazilian Red List may weaken the establishment of conservation measures (Di Dario et al., 2015). Currently, only two national management plans – called National Action Plans – encompass threatened reef species (1. Conservation of Coral Reef Environments and 2. Conservation of Threatened Marine Sharks and Rays) and benefit 45 Brazilian reef fish species (MMA/ICMBio, 2019). These plans aim to mitigate the impacts on Brazilian threatened species and natural ecosystems, setting priorities and guiding conservation actions. Research that assesses and discloses the vulnerability of reef fish species to the government and civil society may assist in the implementation of a greater number of conservation plans in Brazil while raising awareness on nature conservation. Results from this study may help to overcome data gaps that can be used by managers and decision-makers to plan management strategies such as marine spatial planning and fishing restrictions to decrease fishing effort.
Conflicts of interestThe authors declare no conflicts of interest.
C.O.F., M.F.M. and L.S.W. are supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Brazil) scholarships. V.J.G. received a post-doctoral grant #2017/22273-0, São Paulo Research Foundation (FAPESP). This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.